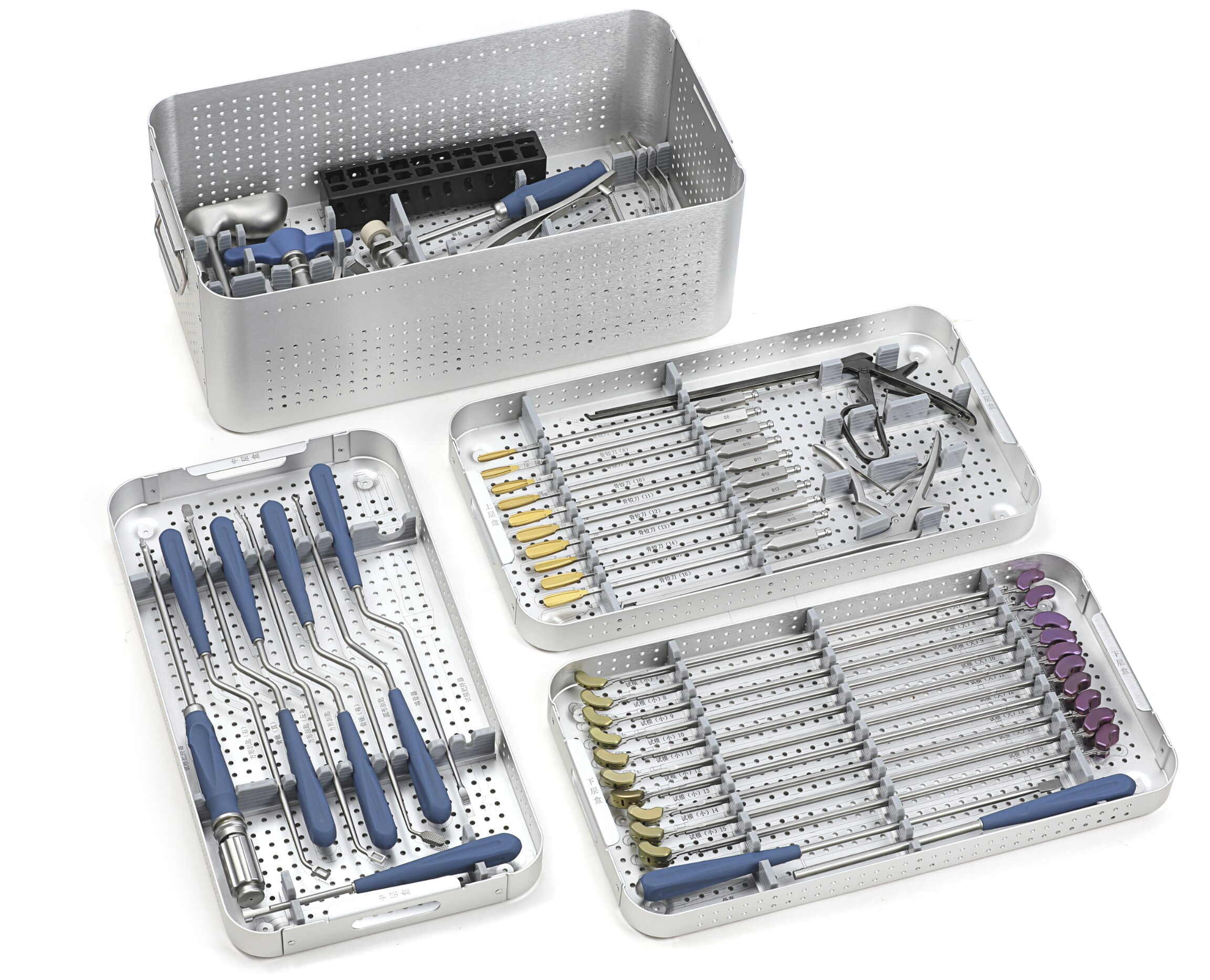
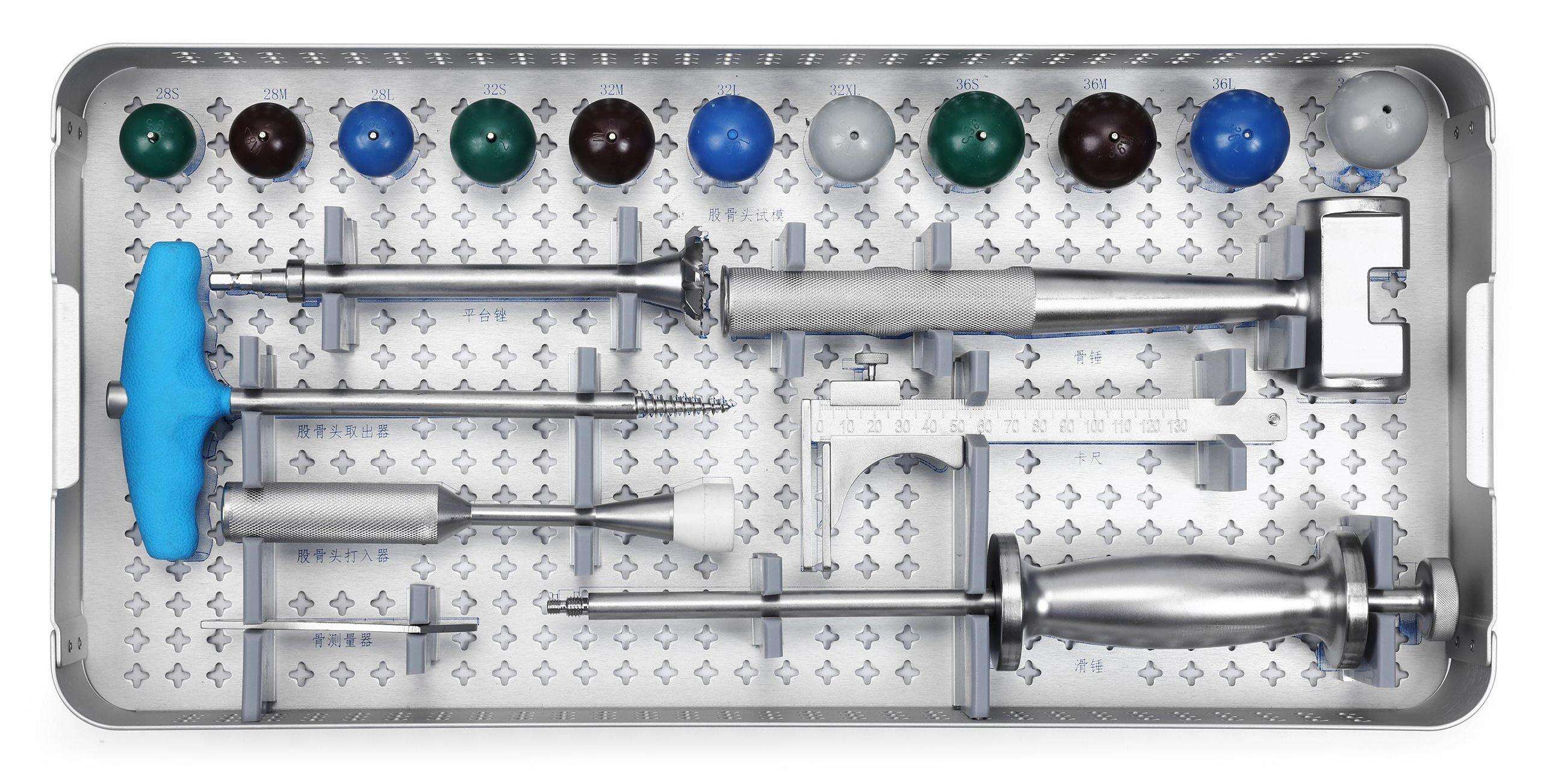
Transforaminal Lumbar Interbody Fusion (TLIF) Instrument Set
Integrated Design. Surgical Precision. OEM/ODM Solutions for Global Spine Partners.
- Overview
- Recommended Products
Product Overview
The Transforaminal Lumbar Interbody Fusion (TLIF) Instrument Set is engineered to support transforaminal access and interbody fusion procedures with accuracy, reproducibility, and safety.
Developed for spine implant brands and medical distributors, this system provides all the essential tools required for disc space preparation, cage insertion, and screw-rod fixation in TLIF surgeries.
Taruk Medical Instruments offers comprehensive OEM/ODM development and manufacturing of TLIF instrument systems.
We help medical brands reduce project complexity by delivering customized, ready-to-assemble instrument kits designed to integrate seamlessly with their existing lumbar fusion implants.
Product Characteristics & Highlights
● Complete System Integration – Instruments designed for disc removal, bone graft placement, and cage insertion in a single modular tray.
● Precision-Guided Design – Angled handles and calibrated depth markings ensure accurate access through the transforaminal route.
● Interchangeable Components – Compatible with various cage geometries and implant designs.
● Enhanced Surgeon Control – Lightweight, ergonomic handles provide stability during decompression and cage delivery.
● Premium Materials – Manufactured from medical-grade stainless steel and titanium with high resistance to corrosion and repeated sterilization.
● Customizable Tray Configuration – Instruments arranged according to client preferences for optimal surgical workflow.
Technical Focus
The TLIF procedure demands controlled access and precise implant positioning within limited visualization.
To meet this challenge, Taruk’s instrument design emphasizes:
● Low-profile access to minimize muscle retraction
● Optimal angulation for cage trajectory
● Reliable mechanical stability under high torsional loads
● Compatibility with different implant manufacturers’ systems
Each instrument undergoes iterative engineering validation to ensure smooth functionality and mechanical endurance.
OEM/ODM Customization Services
As a professional spine surgical instrument manufacturer, Taruk provides flexible OEM/ODM options for TLIF system development:
● Design Customization – Tailored cage inserters, spreaders, and trial instruments per implant dimensions.
● Material Optimization – Selection of medical-grade stainless steel, titanium alloys, or hybrid constructions for weight reduction.
● Prototype & Validation Support – 3D CAD review, prototype sampling, and mechanical testing available for pre-clinical verification.
● Tray & Label Branding – Full customization of trays, engraving, laser-marked logos, and color-coded organization.
● Documentation Assistance – Manufacturing records, inspection reports, and traceability files for CE/FDA submissions.
● Production Scalability – From pilot runs for design evaluation to long-term serial supply.
Through our structured OEM/ODM workflow, clients gain complete visibility — from concept design to validated production.
Manufacturing & Quality Strength
Taruk operates a vertically integrated manufacturing facility with multi-axis CNC centers, polishing and passivation lines, ultrasonic cleaning systems, and controlled packaging zones.
Every process complies with ISO 13485 quality management systems.
Quality checkpoints include:
● Raw material inspection and batch traceability
● Machining accuracy verification via CMM
● Surface finish analysis and passivation quality control
● Functional fit testing using implant dummies
● Packaging integrity validation
We maintain continuous improvement through lean manufacturing and statistical process control (SPC) to ensure long-term product reliability.
Taruk Engineering & Supply Capabilities
● Over two decades of experience in spinal and orthopedic instrument systems
● Expertise in design-for-manufacturability for complex TLIF geometries
● In-house tool validation and mechanical testing lab
● ISO 13485-certified quality and documentation framework
● Global client base in Europe, North America, and Asia-Pacific
Taruk’s advantage lies in combining engineering know-how with precision manufacturing, helping brands shorten development cycles and maintain consistent product performance across batches.
Why Partner with Taruk
● Professional OEM/ODM manufacturer for lumbar fusion instruments
● Transparent communication and technical collaboration at every project phase
● High-precision machining, rapid prototyping, and scalability
● End-to-end support from design to logistics
● Proven record as a reliable supply partner for orthopedic brands worldwide
With Taruk, your TLIF instrument set is not just manufactured — it’s engineered for reliability, consistency, and surgical success.
Inquiry & Collaboration
Looking to develop a customized TLIF Instrument Set that matches your implant systems?
Send us your drawings or specifications, and our engineering team will provide a complete OEM/ODM proposal tailored to your project timeline and market goals.
Taruk Medical Instruments — your dependable OEM/ODM manufacturer for precision spine surgical instrument systems.