The global healthcare industry continues to witness remarkable growth in surgical instrumentation, particularly within the orthopedic sector where trauma cases demand precision-engineered solutions. Manufacturing partnerships between medical device companies and specialized OEM providers have become increasingly critical for delivering high-quality surgical instruments to healthcare facilities worldwide. The orthopedic trauma instruments oem market represents a sophisticated ecosystem where innovation meets regulatory compliance, driving advancements in patient care and surgical outcomes across diverse medical environments.
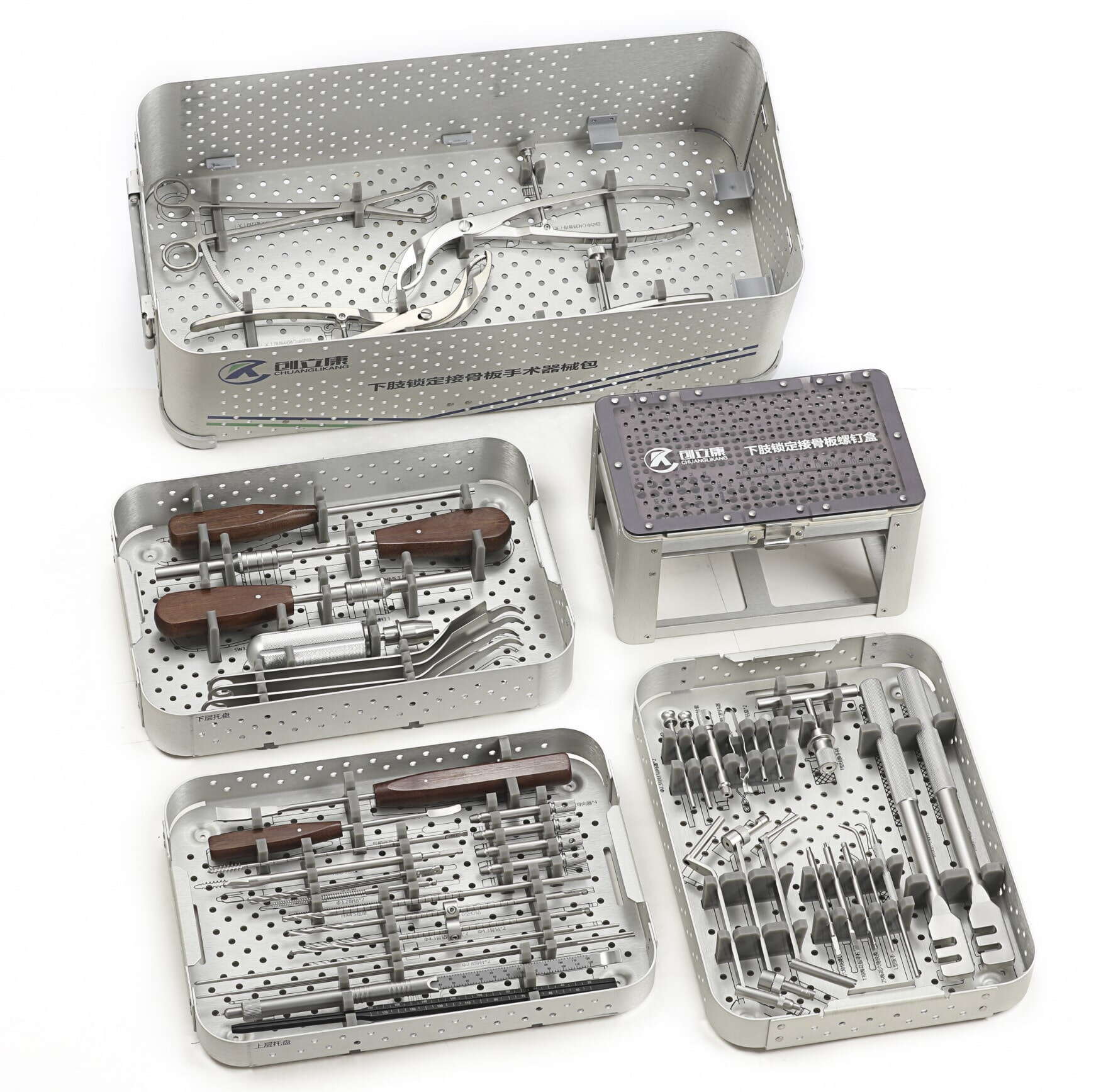
Manufacturing Excellence in Orthopedic Device Production
Quality Standards and Regulatory Compliance
Modern orthopedic trauma instruments oem facilities operate under stringent quality management systems that exceed international medical device standards. These manufacturing environments incorporate advanced metallurgy processes, precision machining capabilities, and comprehensive testing protocols to ensure each instrument meets the exacting requirements of surgical applications. The implementation of ISO 13485 quality management systems alongside FDA compliance frameworks establishes the foundation for reliable, consistent production of trauma instrumentation that surgeons depend on during critical procedures.
Contemporary OEM manufacturing processes integrate sophisticated material science principles with cutting-edge production technologies. Stainless steel alloys, titanium compositions, and specialized coatings undergo rigorous testing to validate biocompatibility, durability, and performance characteristics essential for trauma surgery applications. Quality assurance protocols encompass dimensional accuracy verification, surface finish analysis, and mechanical property validation to guarantee that every instrument delivers optimal functionality throughout its operational lifecycle.
Advanced Manufacturing Technologies
The evolution of manufacturing technologies has transformed how orthopedic trauma instruments oem providers approach product development and production scalability. Computer numerical control machining centers equipped with multi-axis capabilities enable the creation of complex geometries and intricate features that enhance surgical precision and instrument ergonomics. These technological advancements facilitate the production of specialized tools designed for specific trauma procedures, including fracture reduction, bone plating, and intramedullary fixation applications.
Additive manufacturing technologies have emerged as complementary production methods within the orthopedic instrumentation sector. Selective laser melting and electron beam melting processes enable the creation of patient-specific instruments and complex internal geometries that traditional machining cannot achieve. These innovative manufacturing approaches allow OEM providers to offer customized solutions while maintaining cost-effective production capabilities for standard instrument configurations.
Market Dynamics and Global Demand Patterns
Regional Market Expansion
Global demand for orthopedic trauma instruments continues to expand across developed and emerging healthcare markets, driven by aging populations, increased sports-related injuries, and growing access to advanced surgical care. North American and European markets maintain leadership positions in terms of technology adoption and premium instrument demand, while Asian Pacific regions demonstrate rapid growth in volume requirements and local manufacturing capabilities. This geographical diversity creates opportunities for orthopedic trauma instruments oem providers to establish strategic partnerships and expand their global footprint through specialized production facilities.
Emerging markets present unique challenges and opportunities for OEM manufacturers seeking to expand their customer base and production capabilities. Cost-sensitive healthcare systems in developing regions require instruments that maintain clinical effectiveness while meeting aggressive pricing targets. This market dynamic drives innovation in manufacturing efficiency, material optimization, and supply chain management to deliver value-engineered solutions without compromising patient safety or surgical outcomes.
Technology Integration Trends
Digital transformation initiatives within healthcare systems are influencing orthopedic trauma instruments oem development priorities and product design strategies. Smart instrumentation concepts incorporating sensors, connectivity features, and data collection capabilities represent emerging market segments that require specialized manufacturing expertise. These technologically advanced instruments enable real-time surgical feedback, procedure documentation, and post-operative monitoring capabilities that enhance patient care and clinical decision-making processes.
The integration of augmented reality and navigation technologies with traditional trauma instrumentation creates new opportunities for OEM providers to collaborate with software developers and medical device innovators. These partnerships result in hybrid solutions that combine precision mechanical instruments with digital guidance systems, improving surgical accuracy and reducing procedure complexity for healthcare professionals across various skill levels and training backgrounds.
Supply Chain Optimization and Strategic Partnerships
Supplier Relationship Management
Effective supply chain management represents a critical success factor for orthopedic trauma instruments oem operations seeking to maintain competitive advantages in global markets. Strategic partnerships with raw material suppliers, specialized component manufacturers, and logistics providers enable OEM facilities to optimize production costs while ensuring consistent quality and delivery performance. These collaborative relationships facilitate access to advanced materials, innovative manufacturing processes, and technical expertise that enhance product development capabilities and market responsiveness.
Risk mitigation strategies within orthopedic instrumentation supply chains require diversified supplier networks and contingency planning protocols. Geopolitical uncertainties, natural disasters, and pandemic-related disruptions have highlighted the importance of supply chain resilience in maintaining continuous production capabilities. OEM providers increasingly implement multi-sourcing strategies, regional supplier development programs, and inventory optimization systems to minimize operational risks and ensure reliable product availability for their healthcare customers.
Customer Collaboration Models
Modern orthopedic trauma instruments oem relationships extend beyond traditional manufacturing arrangements to encompass collaborative product development, clinical research partnerships, and comprehensive support services. These evolved business models enable OEM providers to work closely with medical device companies, healthcare institutions, and surgical professionals to develop next-generation instruments that address specific clinical challenges and procedural requirements. Such collaborative approaches result in more effective instruments and stronger market positioning for both OEM manufacturers and their customers.
Long-term partnership agreements between OEM manufacturers and medical device companies often include joint investment in research and development activities, shared intellectual property arrangements, and coordinated market expansion strategies. These strategic alliances leverage complementary capabilities and resources to accelerate innovation cycles, reduce development costs, and enhance competitive positioning in rapidly evolving healthcare markets worldwide.
Innovation Drivers and Future Development Pathways
Material Science Advancements
Continuous advancement in material science creates new possibilities for orthopedic trauma instruments oem providers to develop superior products with enhanced performance characteristics. Biocompatible alloys with improved strength-to-weight ratios, corrosion resistance, and fatigue life enable the creation of instruments that deliver superior clinical outcomes while reducing surgeon fatigue during complex procedures. These material innovations often result from collaborative research efforts between OEM manufacturers, material suppliers, and academic institutions focused on advancing surgical instrumentation technology.
Surface modification technologies including specialized coatings, texturing techniques, and antimicrobial treatments represent additional innovation opportunities within the orthopedic instrumentation sector. These advanced surface treatments can improve instrument grip characteristics, reduce bacterial adhesion, and enhance visibility during surgical procedures. OEM providers that invest in these technological capabilities position themselves to offer differentiated products that address specific clinical challenges and regulatory requirements in various global markets.
Sustainability and Environmental Considerations
Environmental sustainability initiatives are increasingly influencing orthopedic trauma instruments oem manufacturing processes and business strategies. Waste reduction programs, energy efficiency improvements, and recycling initiatives help manufacturers reduce their environmental footprint while potentially reducing production costs. These sustainability efforts often align with corporate social responsibility objectives and can enhance brand reputation among environmentally conscious customers and stakeholders.
The development of reusable instrument designs and refurbishment programs represents another approach to sustainability within the orthopedic instrumentation industry. OEM providers that offer comprehensive instrument lifecycle management services, including cleaning protocols, maintenance procedures, and end-of-life recycling programs, create additional value propositions for their customers while supporting environmental stewardship objectives throughout the healthcare supply chain.
Regulatory Landscape and Compliance Strategies
Global Regulatory Harmonization
The evolving regulatory landscape for medical devices continues to shape how orthopedic trauma instruments oem providers approach product development, manufacturing processes, and quality management systems. Regulatory harmonization efforts across major markets facilitate international trade and reduce compliance complexity, while maintaining high safety and efficacy standards for surgical instrumentation. Understanding and adapting to these regulatory requirements represents a critical capability for OEM manufacturers seeking to serve global markets effectively.
Post-market surveillance requirements and adverse event reporting obligations create ongoing compliance responsibilities for orthopedic instrumentation manufacturers. OEM providers must implement comprehensive tracking systems, customer communication protocols, and corrective action procedures to maintain regulatory compliance and protect patient safety. These regulatory compliance capabilities often become competitive differentiators for OEM manufacturers competing for contracts with major medical device companies and healthcare institutions.
Quality Management System Evolution
Modern quality management systems for orthopedic trauma instruments oem operations incorporate risk-based approaches, statistical process control methods, and continuous improvement methodologies. These advanced quality systems enable manufacturers to identify potential issues proactively, optimize production processes, and demonstrate consistent compliance with regulatory requirements across multiple jurisdictions. Investment in robust quality management capabilities supports long-term business growth and customer satisfaction objectives.
Digital quality management platforms integrate manufacturing data, inspection results, and customer feedback to provide comprehensive visibility into product performance and process effectiveness. These technology-enabled quality systems facilitate rapid response to quality issues, support data-driven decision making, and enable predictive quality management approaches that prevent problems before they impact customers or patients. OEM providers that leverage these advanced quality management capabilities often achieve superior operational performance and customer satisfaction levels.
FAQ
What are the key factors to consider when selecting an orthopedic trauma instruments OEM partner
Selecting the right orthopedic trauma instruments oem partner requires careful evaluation of manufacturing capabilities, quality management systems, regulatory compliance history, and technical expertise. Key considerations include ISO certification status, FDA registration, production capacity, material sourcing capabilities, and previous experience with similar instrumentation projects. Additionally, assess the OEM provider's financial stability, customer references, intellectual property protection policies, and long-term partnership commitment to ensure sustainable collaboration.
How do quality standards impact orthopedic trauma instrument manufacturing costs
Quality standards significantly influence manufacturing costs for orthopedic trauma instruments oem operations through requirements for specialized equipment, testing procedures, documentation systems, and skilled personnel. While initial compliance investments may be substantial, effective quality management systems often reduce long-term costs by minimizing defects, preventing recalls, and improving production efficiency. The key is balancing quality requirements with cost optimization strategies to maintain competitive pricing while meeting all regulatory and clinical performance standards.
What role does material selection play in orthopedic instrument performance
Material selection represents a critical factor in orthopedic trauma instruments oem product development, directly impacting instrument durability, biocompatibility, and surgical performance. Stainless steel alloys offer excellent corrosion resistance and cost-effectiveness for many applications, while titanium alloys provide superior strength-to-weight ratios for specialized instruments. Material choice also affects manufacturing processes, surface treatments, and sterilization compatibility, requiring careful consideration of clinical requirements, regulatory standards, and production capabilities.
How are emerging technologies changing orthopedic instrument manufacturing
Emerging technologies are transforming orthopedic trauma instruments oem manufacturing through advanced materials, additive manufacturing processes, and digital integration capabilities. Three-dimensional printing enables rapid prototyping and custom instrument production, while smart manufacturing systems improve quality control and production efficiency. Additionally, digital technologies facilitate remote monitoring, predictive maintenance, and data-driven optimization of manufacturing processes, resulting in improved product quality and reduced production costs for OEM providers and their customers.
Table of Contents
- Manufacturing Excellence in Orthopedic Device Production
- Market Dynamics and Global Demand Patterns
- Supply Chain Optimization and Strategic Partnerships
- Innovation Drivers and Future Development Pathways
- Regulatory Landscape and Compliance Strategies
-
FAQ
- What are the key factors to consider when selecting an orthopedic trauma instruments OEM partner
- How do quality standards impact orthopedic trauma instrument manufacturing costs
- What role does material selection play in orthopedic instrument performance
- How are emerging technologies changing orthopedic instrument manufacturing

