Selecting the right orthopedic device OEM partner represents one of the most critical strategic decisions for medical device companies, distributors, and healthcare organizations seeking to bring high-quality implants and instruments to market. The complexity of orthopedic manufacturing demands not only regulatory compliance and material expertise but also precision engineering capabilities that directly impact patient outcomes. When evaluating potential manufacturing partners, understanding which specific technical competencies separate exceptional manufacturers from adequate ones becomes essential to mitigating supply chain risks, ensuring product quality, and maintaining competitive positioning in an increasingly demanding market.
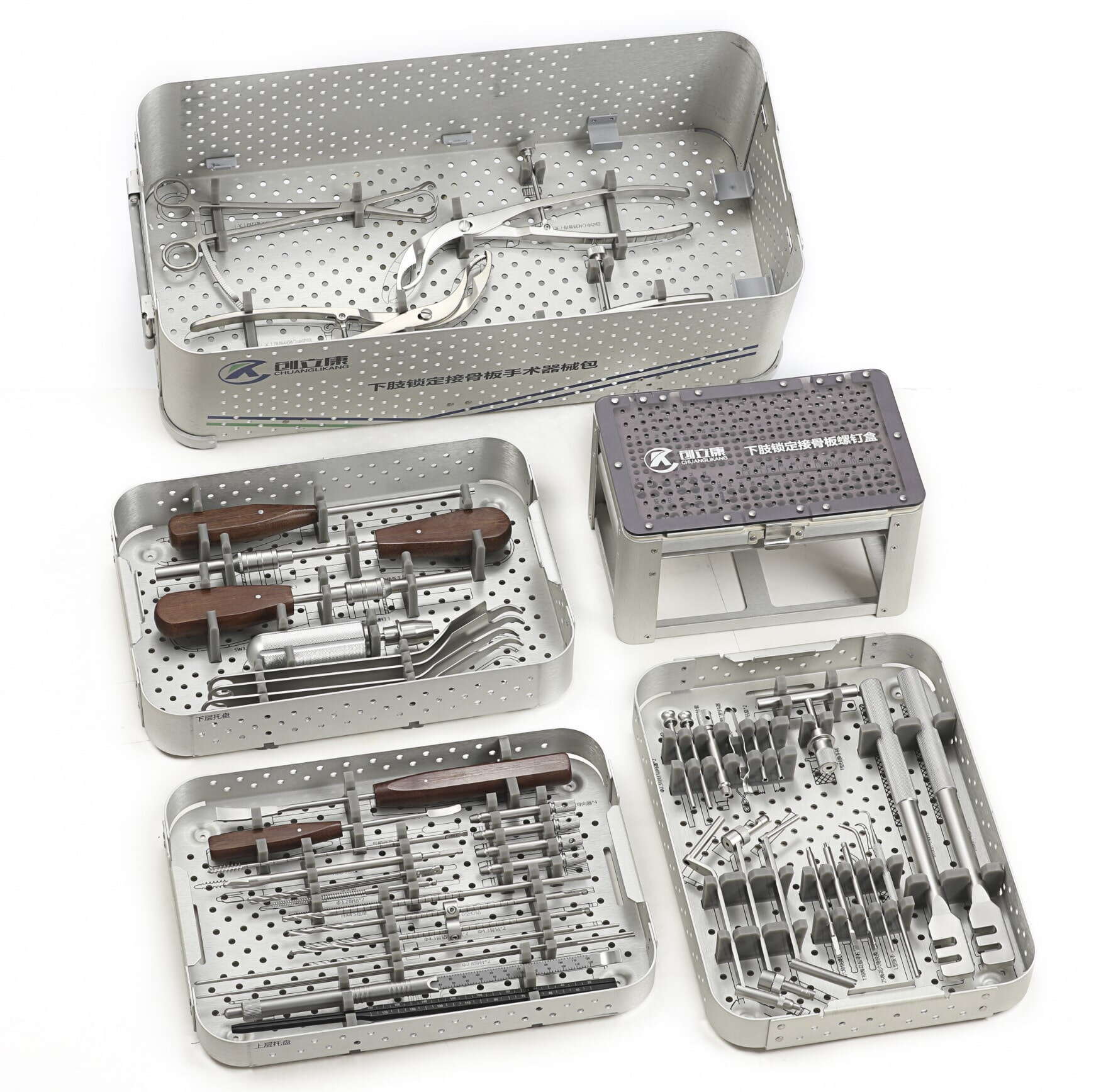
The decision framework for choosing an orthopedic device OEM partner must extend beyond cost considerations and certifications to encompass a comprehensive evaluation of manufacturing capabilities that determine dimensional accuracy, surface finish quality, material integrity, and long-term reliability. This assessment process requires technical depth, as the precision manufacturing requirements for orthopedic implants and surgical instruments far exceed those of general industrial components. From multi-axis CNC machining tolerances measured in microns to surface treatments that influence osseointegration, the technical capabilities of your manufacturing partner directly translate into clinical performance and regulatory approval success. This guide examines seven essential precision manufacturing capabilities that should form the foundation of your partner selection criteria.
Advanced CNC Machining Precision and Tolerance Control
Multi-Axis Machining Capability for Complex Geometries
The foundation of superior orthopedic device OEM manufacturing lies in advanced computer numerical control machining capabilities, particularly five-axis and six-axis CNC systems that enable the production of anatomically complex implant geometries without multiple setups. Leading orthopedic manufacturers invest in high-precision machining centers capable of maintaining tolerances within plus or minus five microns across critical dimensions, which proves essential for components like femoral heads, acetabular cups, and spinal pedicle screws where dimensional accuracy directly affects biomechanical function. When assessing a potential partner, request detailed specifications of their CNC equipment including spindle precision, thermal stability systems, and tool management protocols that ensure consistent dimensional accuracy across production runs.
The capability to machine medical-grade titanium alloys, cobalt-chromium alloys, and stainless steel materials without degradation of tool life or surface integrity indicates substantial manufacturing expertise. Orthopedic device OEM partners with proven multi-axis machining capabilities demonstrate this through documented process validation studies showing consistent achievement of dimensional tolerances, surface roughness specifications, and geometric form tolerances across statistically significant production volumes. Evaluate whether the manufacturer employs in-process measurement systems, climate-controlled machining environments, and predictive maintenance protocols that prevent dimensional drift during extended production campaigns.
Measurement and Quality Verification Systems
Precision machining capabilities remain meaningless without equally sophisticated measurement and verification systems that validate dimensional accuracy throughout production. Leading orthopedic device OEM facilities employ coordinate measuring machines with measurement uncertainty ratios of at least ten-to-one relative to specified tolerances, ensuring that measurement error does not contribute to acceptance of out-of-specification components. Advanced manufacturers integrate optical scanning systems, laser measurement technologies, and statistical process control software that provides real-time feedback to machining operations, enabling immediate correction of dimensional deviations before they affect significant production quantities.
The implementation of comprehensive measurement protocols distinguishes exceptional manufacturing partners from those merely possessing adequate equipment. Request documentation of measurement system analysis studies, gage repeatability and reproducibility data, and calibration protocols that demonstrate metrological traceability to national standards. Manufacturers serving the orthopedic implant industry should maintain temperature-controlled quality laboratories separate from production environments, employ certified quality technicians trained in geometric dimensioning and tolerancing interpretation, and utilize specialized fixtures that ensure repeatable component orientation during measurement activities.
Surface Finish Control and Characterization
Surface finish characteristics profoundly influence both the functional performance and biological response to orthopedic implants, making surface finish control a critical manufacturing capability. Top-tier orthopedic device OEM partners demonstrate expertise in achieving specified surface roughness parameters through optimized cutting strategies, appropriate tool selection, and validated finishing processes. The capability to produce mirror-polished articulating surfaces with roughness values below fifty nanometers Ra alongside textured bony-contact surfaces with controlled micro-roughness demonstrates manufacturing versatility essential for comprehensive implant portfolios.
Assessment of surface finish capabilities should examine both the measurement technologies employed and the process control methods that ensure consistency. Manufacturers should utilize white light interferometry, confocal microscopy, or contact profilometry systems capable of characterizing three-dimensional surface topography beyond simple roughness averages. Documentation of surface finish validation studies across different materials, geometries, and machining strategies provides confidence in the manufacturer's ability to consistently meet specifications that directly affect wear resistance, friction characteristics, and osseointegration potential in clinical applications.
Material Processing Expertise and Traceability
Medical-Grade Material Sourcing and Certification
The selection and processing of raw materials forms the foundation of implant quality, making material expertise a non-negotiable capability for any orthopedic device OEM partner. Leading manufacturers maintain established relationships with certified material suppliers who provide mill certificates, chemical composition analyses, and mechanical property documentation traceable to specific heat lots. The capability to source and verify titanium alloys conforming to ASTM F136 and F1472 standards, cobalt-chromium alloys meeting ASTM F75 and F1537 specifications, and medical-grade stainless steels compliant with ASTM F138 requirements demonstrates the supply chain maturity necessary for regulated medical device production.
Material traceability systems that link finished devices back to original material certifications represent essential infrastructure for regulatory compliance and quality investigation capabilities. Evaluate whether prospective manufacturing partners employ enterprise resource planning systems that maintain complete material genealogy, implement first-in-first-out inventory management to prevent material aging issues, and conduct incoming material inspections that verify chemical composition and mechanical properties before materials enter production. Manufacturers who invest in spectroscopy equipment for material verification and maintain segregated storage for different material grades demonstrate the operational discipline necessary for reliable orthopedic device production.
Heat Treatment and Material Property Optimization
Controlled heat treatment processes critically influence the mechanical properties, microstructure, and corrosion resistance of orthopedic implant materials, making thermal processing expertise a key differentiator among orthopedic device OEM candidates. Superior manufacturers operate vacuum furnaces, atmosphere-controlled ovens, and solution annealing equipment with documented temperature uniformity studies and calibrated instrumentation that ensures precise thermal cycles. The capability to perform stress relief treatments, solution annealing, aging treatments, and specialized thermal processes tailored to specific alloy systems demonstrates metallurgical sophistication beyond basic machining capabilities.
Assessment of heat treatment capabilities should include review of furnace qualification documentation, process validation studies demonstrating consistent achievement of specified mechanical properties, and microstructural analysis capabilities that verify appropriate grain structure and phase composition. Leading manufacturers employ hardness testing, tensile testing, and metallographic examination as routine verification methods, maintaining statistical process control charts that track mechanical property trends across production campaigns. The integration of heat treatment operations within the same facility as machining operations typically provides better process control and traceability compared to reliance on external service providers.
Passivation and Surface Treatment Protocols
Chemical passivation treatments that enhance corrosion resistance represent essential final processing steps for stainless steel and titanium orthopedic components. Accomplished orthopedic device OEM partners maintain validated passivation protocols conforming to ASTM F86 and ASTM B600 standards, employing chemical analysis and process monitoring that ensures consistent treatment effectiveness. The capability to perform nitric acid passivation, electropolishing, and specialized surface treatments while maintaining dimensional integrity and surface finish specifications demonstrates process control sophistication necessary for implantable device production.
Evaluation of surface treatment capabilities should examine both the chemical processing infrastructure and the analytical methods used to verify treatment effectiveness. Manufacturers should perform copper sulfate testing, salt spray exposure studies, and electrochemical corrosion testing that confirms enhanced corrosion resistance following passivation. Documentation of water quality monitoring, chemical bath analysis, and process parameter control provides confidence in treatment consistency across production volumes. Integration of passivation operations with appropriate pre-cleaning and post-treatment rinsing procedures prevents contamination that could compromise implant biocompatibility.
Coating Technologies and Surface Modification Capabilities
Plasma Spray and Thermal Coating Processes
Advanced surface coating technologies enable orthopedic implants to achieve biological fixation and enhanced wear resistance that cannot be obtained through substrate materials alone. Leading orthopedic device OEM manufacturers operate plasma spray systems, high-velocity oxy-fuel coating equipment, and specialized thermal spray technologies capable of depositing hydroxyapatite, titanium, and porous metal coatings with controlled thickness, porosity, and bond strength. The capability to apply coatings that meet ISO 13779 standards for hydroxyapatite coatings and FDA guidance for porous coatings demonstrates technical sophistication essential for modern implant design requirements.
Assessment of coating capabilities should include examination of coating parameter documentation, cross-sectional analysis showing coating microstructure and substrate bonding, and mechanical testing data demonstrating adhesion strength and coating cohesion. Superior manufacturers employ automated coating systems with real-time monitoring of spray parameters including powder feed rate, plasma gas composition, substrate temperature, and spray distance. The integration of coating operations with subsequent quality verification including thickness measurement, porosity characterization, and stereo microscopy examination ensures coating consistency critical for osseointegration performance and long-term implant stability.
Physical Vapor Deposition and Advanced Coatings
Physical vapor deposition technologies including ion beam assisted deposition and magnetron sputtering enable ultra-thin, highly adherent coatings that enhance wear resistance and reduce ion release from orthopedic implant surfaces. Manufacturers offering PVD capabilities can apply titanium nitride, zirconium nitride, and diamond-like carbon coatings with thickness uniformity and surface conformality impossible to achieve through thermal spray methods. These advanced coating options provide design flexibility for articulating surfaces subject to high contact stresses and tribological challenges where conventional coating technologies prove inadequate.
Evaluation of PVD capabilities should assess vacuum system performance, coating uniformity across complex geometries, and validated process parameters that ensure coating adhesion and wear resistance. Leading manufacturers employ scratch testing, nano-indentation hardness measurement, and accelerated wear testing that quantifies coating performance under simulated physiological conditions. Documentation of coating process validation, including design of experiments studies correlating process parameters with coating properties, demonstrates the scientific approach necessary for reliable coating application that meets demanding orthopedic performance requirements.
Surface Texturing and Modification Technologies
Controlled surface texturing through chemical etching, grit blasting, laser machining, and electrochemical processes enables creation of micro-scale and nano-scale topographies that influence cellular response and osseointegration. Accomplished orthopedic device OEM facilities employ multiple surface modification technologies, allowing selection of optimal surface characteristics for different implant regions and biological interface requirements. The capability to produce uniformly roughened surfaces with controlled texture parameters, create graduated surface features that transition between implant regions, and apply localized surface treatments to specific component areas demonstrates manufacturing flexibility that supports innovative implant designs.
Assessment should examine the range of available surface modification technologies, the process control methods employed to ensure texture consistency, and the characterization techniques used to verify surface topography. Manufacturers should provide scanning electron microscopy images documenting surface morphology, quantitative surface roughness data across multiple length scales, and biological response data correlating surface features with cellular behavior. Integration of surface modification capabilities with computer-aided design systems that specify surface characteristics as engineered features rather than post-processing variables indicates advanced manufacturing maturity beneficial for complex product development initiatives.
Assembly and Instrumentation Manufacturing Competencies
Precision Assembly and Component Integration
Many orthopedic systems comprise multiple components requiring precise assembly, creating dependencies on assembly capabilities beyond individual component manufacturing. Superior orthopedic device OEM partners demonstrate expertise in modular implant assembly, instrument tray configuration, and multi-component system integration that maintains specified tolerances and functional performance. The capability to perform press-fitting operations with controlled interference, apply medical-grade adhesives with validated bonding protocols, and execute mechanical locking mechanisms that withstand specified separation forces represents essential competency for comprehensive product portfolios including modular hip stems, polyaxial spinal systems, and assembled instrument sets.
Evaluation of assembly capabilities should examine documented assembly procedures, torque control and verification systems, and functional testing protocols that confirm assembled product performance. Leading manufacturers employ poka-yoke error-proofing devices, vision inspection systems that verify correct component orientation and placement, and statistical sampling plans that ensure assembly quality across production volumes. The integration of assembly operations within clean room environments appropriate for medical device manufacturing, combined with documented assembly validation studies demonstrating process capability, provides confidence in consistent product quality for complex multi-component orthopedic systems.
Surgical Instrument Manufacturing Expertise
Orthopedic surgical instrument manufacturing demands precision tolerances, ergonomic design execution, and durability under repeated sterilization cycles that distinguish instrument production from implant manufacturing. Accomplished orthopedic device OEM facilities demonstrate competency across instrument categories including cutting guides, drill bits, impactors, extractors, and specialized insertion tools. The capability to produce instruments meeting dimensional accuracy requirements while withstanding autoclave sterilization, chemical disinfection, and mechanical stresses encountered during surgical procedures requires material selection expertise, heat treatment knowledge, and finishing processes distinct from implant production requirements.
Assessment of instrument manufacturing capabilities should include examination of dimensional verification protocols specific to functional requirements, durability testing documentation showing instrument performance across simulated use cycles, and corrosion resistance data demonstrating survival through specified sterilization protocols. Superior manufacturers maintain separate production areas for reusable instruments versus implantable components, employ specialized grinding and lapping processes that achieve sharp cutting edges with specified geometry, and conduct functional testing that verifies instrument performance under clinically representative loading conditions before product release.
Custom Instrument Tray Design and Manufacturing
Comprehensive orthopedic device OEM partnerships often encompass custom instrument tray design and manufacturing that protects surgical instruments during sterilization and facilitates efficient surgical workflow. Leading manufacturers offer capabilities spanning instrument tray engineering, silicone foam insert fabrication, aluminum or stainless steel tray manufacturing, and laser engraving for permanent instrument identification. The ability to design tray systems optimized for specific sterilization methods, create custom foam configurations that secure instruments and prevent damage during handling, and provide integrated labeling solutions that maintain traceability throughout the product lifecycle demonstrates full-service capabilities valuable for market-ready product delivery.
Evaluation should assess the manufacturer's design capabilities including computer-aided tray layout optimization, foam material selection appropriate for repeated sterilization exposure, and tray structural design that withstands stacking loads and handling stresses. Manufacturers should provide sterilization validation data confirming tray compatibility with steam sterilization, hydrogen peroxide plasma sterilization, and ethylene oxide sterilization as appropriate for intended use. Integration of instrument tray manufacturing with instrument and implant production within a single facility typically provides better coordination, shorter lead times, and simplified regulatory documentation compared to sourcing tray systems from separate suppliers.
Quality Systems and Regulatory Compliance Infrastructure
ISO 13485 and FDA Registration Compliance
Robust quality management systems certified to ISO 13485 medical device standards represent foundational requirements for any orthopedic device OEM partner serving regulated markets. Superior manufacturers maintain current ISO 13485 certification with documented quality manual, controlled procedures, and objective evidence of system effectiveness through internal audits and management reviews. Beyond basic certification, evaluation should assess the maturity and effectiveness of the quality system through examination of corrective action and preventive action records, nonconformance trending analysis, and supplier management programs that extend quality requirements throughout the supply chain.
For manufacturers serving United States markets, FDA establishment registration and adherence to Quality System Regulation requirements under 21 CFR Part 820 constitute non-negotiable prerequisites. Assessment should verify current FDA registration status, review FDA inspection history including any Form 483 observations and subsequent corrective actions, and examine design control documentation demonstrating systematic product development processes. Manufacturers with established FDA compliance track records, documented design history files, and experience supporting 510k or PMA regulatory submissions provide substantial risk mitigation compared to partners lacking regulatory infrastructure or North American market experience.
Process Validation and Statistical Process Control
Comprehensive process validation documentation demonstrating that manufacturing processes consistently produce devices meeting predetermined specifications represents essential evidence of manufacturing capability and regulatory compliance. Leading orthopedic device OEM partners maintain installation qualification, operational qualification, and performance qualification protocols for critical manufacturing equipment including CNC machines, coating systems, and sterilization equipment. The implementation of statistical process control across critical-to-quality parameters with documented control limits, capability indices, and trending analysis demonstrates proactive quality management rather than reactive inspection-based approaches.
Evaluation should examine specific validation protocols, statistical analysis methods employed, and revalidation procedures triggered by process changes or equipment modifications. Superior manufacturers employ design of experiments methodologies that establish scientifically-based process parameters, maintain process capability databases that track performance over extended periods, and implement continuous improvement programs that optimize processes based on statistical analysis. The integration of statistical process control with manufacturing execution systems that provide real-time visibility into process performance indicates advanced manufacturing maturity beneficial for consistent product quality and efficient production scale-up.
Traceability Systems and Documentation Control
Comprehensive traceability from raw materials through finished device distribution enables effective recalls, quality investigations, and regulatory compliance verification. Accomplished orthopedic device OEM facilities implement serialization systems, lot tracking protocols, and document management infrastructure that maintains complete device history records as required by regulatory frameworks. The capability to trace any finished device back to specific material lots, processing equipment, operator qualifications, and inspection results within minutes rather than days demonstrates information system maturity essential for modern medical device manufacturing.
Assessment should evaluate the enterprise systems employed for traceability including material requirements planning software, quality management information systems, and manufacturing execution platforms. Leading manufacturers employ barcode scanning, radio-frequency identification, or other automated data collection methods that minimize manual documentation errors and provide real-time visibility into production status. Integration of traceability systems with customer complaint handling, corrective action management, and post-market surveillance activities demonstrates systemic thinking about quality management extending beyond manufacturing operations into product lifecycle management.
Engineering Support and Product Development Collaboration
Design for Manufacturability Expertise
Superior orthopedic device OEM partnerships extend beyond contract manufacturing to encompass collaborative engineering support that optimizes product designs for efficient, reliable manufacturing. Leading manufacturers employ design engineers with orthopedic domain knowledge who provide design for manufacturability feedback addressing tolerance stack-up analysis, material selection optimization, manufacturing process selection, and cost reduction opportunities. The availability of engineering resources capable of conducting finite element analysis, tolerance analysis, and manufacturing simulation provides substantial value during product development phases, preventing costly design iterations and accelerating time-to-market.
Evaluation should assess the engineering staff size and qualifications, review examples of design optimization projects completed for other clients, and discuss the manufacturer's approach to new product introduction. Manufacturers offering concurrent engineering support with regular design review meetings, documented design input and output records, and collaborative problem-solving during development phases typically deliver better outcomes than purely transactional manufacturing relationships. The willingness to sign non-disclosure agreements, establish clear intellectual property provisions, and commit engineering resources to support development activities indicates partnership orientation rather than purely service-provider mentality.
Prototype and Low-Volume Production Capabilities
Flexible manufacturing capabilities supporting both prototype production and full-scale manufacturing within the same facility provide significant advantages for product development timelines and design validation activities. Accomplished orthopedic device OEM partners demonstrate rapid prototyping capabilities using production-equivalent materials and processes, enabling design verification testing with components representative of commercial manufacturing. The ability to efficiently transition from prototype quantities through design validation builds and into commercial production without changing manufacturing facilities or processes reduces technical risk and accelerates regulatory submission timelines.
Assessment should examine the manufacturer's capacity allocation between prototype and production activities, typical lead times for prototype component delivery, and documented examples of successful transitions from development through commercial launch. Superior manufacturers maintain dedicated prototype manufacturing resources including quick-change tooling systems, flexible CNC programming approaches, and quality verification protocols scaled appropriately for small quantities while maintaining production-representative processes. The integration of prototype capabilities with engineering support services including design iteration, test sample production, and process optimization studies provides comprehensive development support valuable for emerging orthopedic device companies and established organizations launching innovative products.
Regulatory Documentation Support and Technical File Development
Comprehensive regulatory submission documentation including device master records, design history files, and technical documentation supporting CE marking or FDA clearance requires substantial technical writing expertise and regulatory knowledge. Leading orthopedic device OEM manufacturers provide regulatory support services including compilation of manufacturing process descriptions, preparation of material and biocompatibility documentation, and development of test reports supporting regulatory submissions. The availability of regulatory affairs specialists familiar with orthopedic device requirements, international regulatory frameworks, and specific submission formats reduces client burden and accelerates approval timelines.
Evaluation should assess the manufacturer's experience supporting regulatory submissions across different jurisdictions, review examples of regulatory documentation prepared for other clients, and discuss the division of responsibilities for regulatory activities. Manufacturers offering turnkey regulatory support including clinical evaluation report preparation, risk analysis documentation, and post-market surveillance planning provide substantial value beyond manufacturing services. The willingness to serve as authorized representative or legal manufacturer for specific markets, combined with established quality agreements and design control procedures, enables smaller organizations to access regulated markets without establishing complete in-house regulatory infrastructure.
FAQ
What certifications should an orthopedic device OEM partner hold to ensure regulatory compliance?
An orthopedic device OEM partner must maintain ISO 13485 certification for medical device quality management as a fundamental requirement for serving regulated markets. For United States distribution, the manufacturer requires FDA establishment registration and demonstrated compliance with 21 CFR Part 820 Quality System Regulation. European market access necessitates compliance with Medical Device Regulation (MDR) requirements including technical documentation and post-market surveillance systems. Additional beneficial certifications include ISO 9001 for general quality management and industry-specific accreditations such as Nadcap for specialized processes. Beyond certificates, examine actual audit records, regulatory inspection outcomes, and quality system effectiveness through metrics like on-time delivery, defect rates, and corrective action closure timelines.
How can I verify that an orthopedic device OEM has adequate precision manufacturing capabilities?
Verification of precision manufacturing capabilities requires on-site facility audits examining actual production equipment, measurement systems, and quality records rather than relying solely on marketing materials. Request equipment lists with specifications including CNC machine tolerances, coordinate measuring machine uncertainties, and surface finish measurement capabilities. Review process validation documentation showing statistical evidence that manufacturing processes consistently achieve specified tolerances across multiple production runs. Examine first article inspection reports, process capability studies with calculated Cpk values, and measurement system analysis data demonstrating adequate measurement precision. Request sample components for independent dimensional verification and metallurgical analysis. Leading manufacturers welcome technical audits and provide transparent access to quality records, equipment specifications, and process documentation supporting their claimed capabilities.
What differentiates a basic contract manufacturer from a true orthopedic device OEM partnership?
A true orthopedic device OEM partnership extends beyond transactional manufacturing services to encompass collaborative engineering support, design optimization input, and shared commitment to product success. Strategic OEM partners provide design for manufacturability feedback during development phases, offer prototype and pilot production capabilities using production-equivalent processes, and maintain flexible capacity allocation supporting both development and commercial needs. Partnership-oriented manufacturers invest in understanding client product strategies, maintain confidential intellectual property protections, and provide proactive communication regarding quality issues, supply chain challenges, or improvement opportunities. Financial stability, long-term facility investment, and demonstrated client retention rates indicate partnership commitment beyond project-based relationships. The availability of dedicated account management, regular business reviews, and willingness to establish strategic agreements with volume commitments distinguishes partnership relationships from simple supplier transactions.
How important is geographic proximity when selecting an orthopedic device OEM partner?
Geographic proximity offers advantages including easier facility audits, simplified communication across time zones, reduced shipping costs, and faster response times for quality issues or engineering support requirements. However, geographic location should be evaluated within the context of total capability, quality track record, and value proposition rather than as the primary selection criterion. Manufacturers located in established medical device clusters often provide access to specialized supply chains, experienced technical talent, and regulatory expertise concentrated in those regions. For companies requiring frequent design iterations, prototype production, or hands-on collaboration during development phases, regional proximity provides substantial workflow benefits. Conversely, established products in commercial production with stable specifications may benefit less from proximity advantages. Consider regulatory environment, intellectual property protection, export control requirements, and total landed cost when evaluating domestic versus international manufacturing partners.
Table of Contents
- Advanced CNC Machining Precision and Tolerance Control
- Material Processing Expertise and Traceability
- Coating Technologies and Surface Modification Capabilities
- Assembly and Instrumentation Manufacturing Competencies
- Quality Systems and Regulatory Compliance Infrastructure
- Engineering Support and Product Development Collaboration
-
FAQ
- What certifications should an orthopedic device OEM partner hold to ensure regulatory compliance?
- How can I verify that an orthopedic device OEM has adequate precision manufacturing capabilities?
- What differentiates a basic contract manufacturer from a true orthopedic device OEM partnership?
- How important is geographic proximity when selecting an orthopedic device OEM partner?

