The orthopedic device industry operates at the intersection of life-saving medical intervention and precision engineering, where even microscopic deviations can compromise patient outcomes. For surgeons relying on implants and instruments during critical procedures, the integrity of every component is paramount. This reality places extraordinary demands on orthopedic OEM partners, who must navigate a complex landscape where material science, manufacturing excellence, and regulatory adherence converge. Understanding why these three pillars are non-negotiable is essential for healthcare institutions, medical device companies, and procurement professionals seeking reliable manufacturing partnerships in this high-stakes sector.
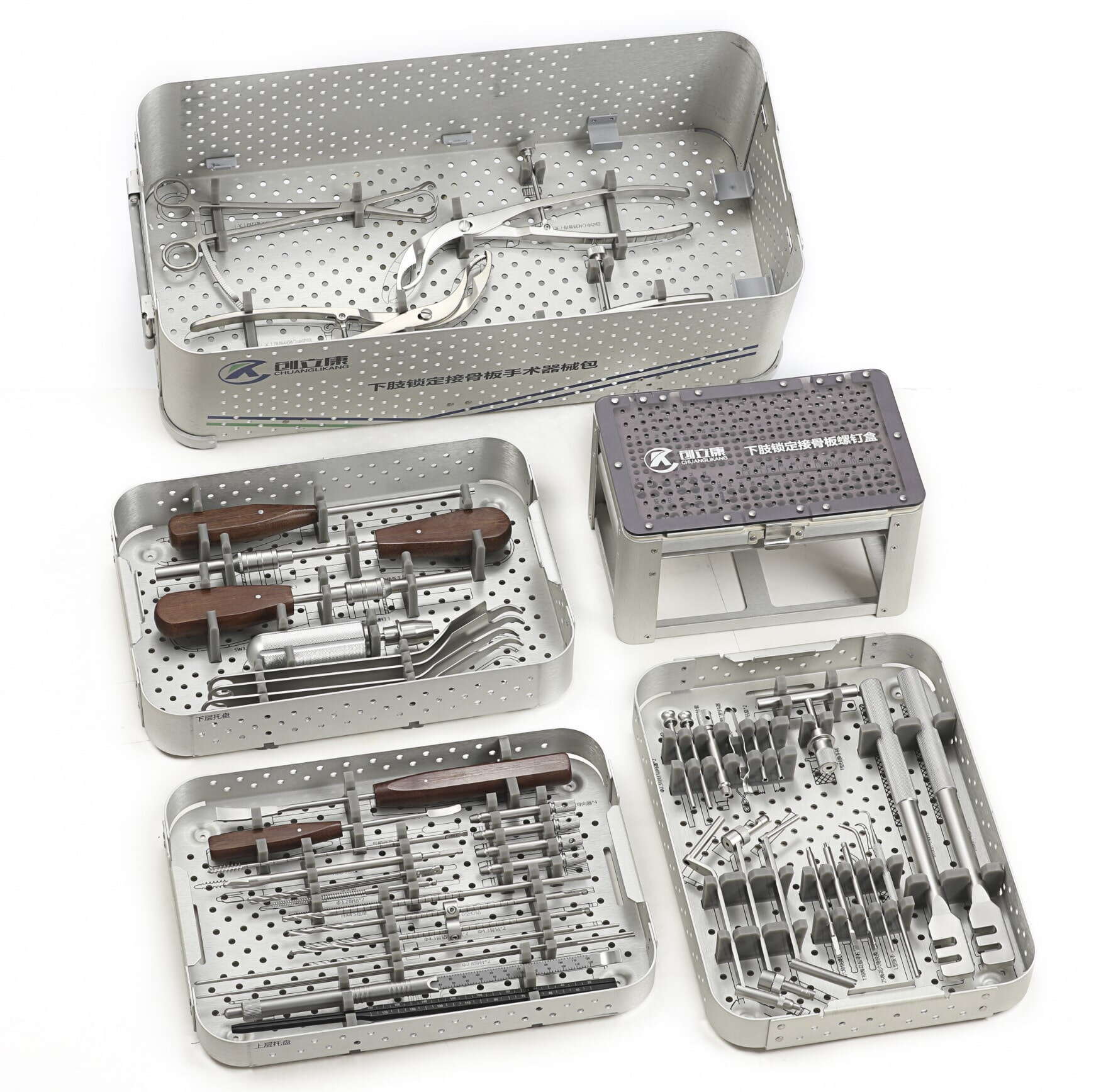
The question of why material selection, process control, and compliance cannot be compromised in orthopedic OEM manufacturing stems from the fundamental nature of these devices. Unlike consumer products where performance tolerances allow greater flexibility, orthopedic implants and instruments become integrated into human anatomy or facilitate surgical procedures that permanently alter skeletal structures. A substandard material choice can trigger immune responses, a flawed manufacturing process can create stress concentrations leading to catastrophic implant failure, and regulatory non-compliance can halt product distribution while exposing patients to unvalidated risks. For orthopedic OEM providers, these three elements form an inseparable triad that determines not only commercial viability but also ethical responsibility to end users.
The Critical Role of Material Selection in Orthopedic OEM Manufacturing
Biocompatibility Requirements That Define Material Viability
Material selection in orthopedic OEM manufacturing begins with biocompatibility assessment, a rigorous evaluation process that determines whether a substance can coexist with living tissue without triggering adverse reactions. Titanium alloys, stainless steel variants, and cobalt-chromium compositions dominate this space because they have demonstrated long-term compatibility with bone and soft tissue across decades of clinical use. However, biocompatibility is not a binary characteristic but rather a spectrum influenced by alloy purity, surface finishing, and the specific anatomical location where the device will be implanted. An orthopedic OEM manufacturer must maintain comprehensive documentation proving that selected materials meet ISO 10993 standards, which outline biological evaluation protocols for medical devices intended for human contact.
The complexity intensifies when considering that different anatomical sites present distinct biochemical environments. Materials suitable for load-bearing femoral stems face different corrosion challenges than those used in craniofacial reconstruction plates. Orthopedic OEM partners must therefore maintain material databases that account for pH variations, ionic concentrations, and mechanical stress patterns specific to each application. This expertise extends beyond basic material science to include understanding how surface treatments like plasma spraying or anodization affect tissue integration, and how these modifications might alter the material's fundamental biocompatibility profile over the device's intended lifespan.
Mechanical Performance Properties That Ensure Device Longevity
Beyond biocompatibility, mechanical properties determine whether an orthopedic device can withstand the extraordinary forces generated during normal physiological activity. Hip and knee implants routinely experience cyclic loading patterns that, over a typical patient lifespan, may exceed millions of repetitions. An orthopedic OEM manufacturer must select materials with fatigue resistance that prevents crack initiation and propagation under these demanding conditions. Tensile strength, yield strength, and elastic modulus must align with the specific application, ensuring that implants provide adequate support without stress-shielding adjacent bone tissue, which can lead to bone resorption and implant loosening.
Material selection also impacts the machinability and formability characteristics that influence manufacturing efficiency and precision. Some high-performance alloys offer superior mechanical properties but present significant challenges during CNC machining or forging operations. A skilled orthopedic OEM provider balances these considerations, selecting materials that meet clinical performance requirements while remaining compatible with available manufacturing capabilities. This decision-making process requires deep understanding of material behavior during various fabrication steps, including how work hardening affects subsequent machining operations or how heat treatment cycles influence grain structure and final mechanical properties.
Traceability Standards That Protect Supply Chain Integrity
Material traceability represents a non-negotiable requirement in orthopedic OEM manufacturing, establishing an unbroken documentation chain from raw material certification through final device implantation. Each batch of medical-grade alloy arrives with mill certificates documenting chemical composition, mechanical testing results, and processing history. Reputable orthopedic OEM manufacturers implement lot tracking systems that link these certificates to specific production runs, enabling rapid identification and isolation if material defects are discovered post-distribution. This capability becomes critical during adverse event investigations, where regulators may require proof that implanted devices were manufactured from certified materials meeting specified standards.
The traceability imperative extends to understanding material sourcing geography and supply chain vulnerability. Certain alloying elements critical to orthopedic applications face geopolitical supply constraints, making dual-sourcing strategies essential for business continuity. An experienced Orthopedic OEM partner maintains relationships with multiple certified suppliers while ensuring that material substitutions undergo full validation testing before introduction into production. This redundancy protects medical device companies from supply disruptions that could compromise product availability and patient care continuity.
Process Control as the Foundation of Manufacturing Reliability
Precision Machining Tolerances That Enable Surgical Predictability
Manufacturing process control in orthopedic OEM operations centers on achieving dimensional tolerances that often reach single-digit micron levels. These specifications are not arbitrary engineering preferences but direct requirements for device functionality and surgical technique compatibility. Locking compression plates, for example, depend on precise thread geometries that create predictable angular stability when screws engage the plate holes. Deviations of even fifty microns in thread pitch or angle can compromise this mechanical interlock, reducing fixation strength and potentially leading to construct failure during the healing process.
Achieving these tolerances requires advanced CNC machining centers equipped with real-time measurement systems and environmental controls that compensate for thermal expansion effects. Orthopedic OEM facilities invest in five-axis machining capabilities that minimize part repositioning, reducing cumulative positioning errors that compound across multiple setups. Tool wear monitoring systems automatically compensate for gradual dimensional drift, ensuring that the thousandth part in a production run maintains the same geometric fidelity as the first. This level of process sophistication represents a substantial capital investment that separates competent orthopedic OEM providers from those attempting to serve this market with general manufacturing capabilities.
Surface Finishing Techniques That Influence Biological Response
Surface characteristics profoundly influence how orthopedic devices interact with surrounding tissue, making surface finishing a critical process control parameter rather than a cosmetic consideration. Roughness values measured in micrometers determine whether bone cells can effectively adhere to implant surfaces, with specific roughness ranges promoting osseointegration while smoother finishes minimize bacterial adhesion on articulating surfaces. Orthopedic OEM manufacturers employ multiple finishing techniques including electropolishing, bead blasting, and chemical etching, each producing distinct surface topographies optimized for specific device functions and anatomical locations.
Process control in surface finishing extends beyond achieving target roughness values to include contamination prevention and residue elimination. Even trace quantities of polishing compounds, cleaning solvents, or handling residues can trigger inflammatory responses or interfere with subsequent coating processes. Leading orthopedic OEM facilities implement cleanroom protocols during finishing operations, use pharmaceutical-grade cleaning agents, and validate residue levels through surface analysis techniques like X-ray photoelectron spectroscopy. These measures ensure that finished surfaces meet not only dimensional specifications but also cleanliness standards essential for biocompatibility and coating adhesion.
Statistical Process Control Systems That Prevent Defect Propagation
Modern orthopedic OEM manufacturing relies on statistical process control methodologies that identify process drift before it produces out-of-specification parts. By continuously monitoring critical dimensions, force values, and process parameters, manufacturers establish control charts that reveal patterns indicating tool wear, machine calibration drift, or material property variations. This proactive approach enables corrective action during production rather than discovering defects during final inspection, reducing scrap rates and protecting delivery schedules while ensuring consistent product quality.
Implementation of robust statistical process control requires both technological infrastructure and cultural commitment to data-driven decision making. Orthopedic OEM facilities deploy coordinate measuring machines integrated with production management systems that automatically flag measurements approaching specification limits. Quality engineers analyze control chart patterns to distinguish between common cause variation inherent to the process and special cause variation requiring intervention. This analytical capability transforms quality control from a reactive inspection function into a predictive manufacturing optimization tool, enabling continuous improvement in process capability and product consistency.
Regulatory Compliance as a Market Access Imperative
Quality Management System Requirements That Govern Operations
Regulatory compliance in orthopedic OEM manufacturing begins with implementation of quality management systems that meet ISO 13485 standards, the internationally recognized framework for medical device manufacturing. This comprehensive system governs every aspect of operations from design control through post-market surveillance, establishing documented procedures that ensure consistent execution of critical activities. For orthopedic OEM providers, ISO 13485 certification is not optional marketing credential but rather a prerequisite for market participation, as regulatory authorities worldwide reference these standards when evaluating manufacturing facility suitability.
Quality management system maintenance requires ongoing commitment extending beyond initial certification. Orthopedic OEM facilities conduct regular internal audits verifying procedure adherence, document control effectiveness, and corrective action implementation. Management review meetings examine quality metrics, customer complaints, and regulatory developments to ensure the quality system evolves with changing requirements and business conditions. This systematic approach creates institutional discipline that prevents the gradual process degradation that can occur when quality depends on individual vigilance rather than systemic controls.
Design History File Documentation That Proves Device Safety
For orthopedic OEM manufacturers engaged in custom device development or design modification activities, design history file documentation provides the evidentiary foundation demonstrating that devices are safe and effective for their intended use. This comprehensive record encompasses design inputs derived from clinical needs, design outputs including engineering drawings and specifications, design verification testing proving the design meets requirements, and design validation studies confirming the device performs as intended in simulated use conditions. Regulatory reviewers scrutinize these files during premarket submission evaluations and facility inspections, making documentation completeness and technical rigor essential for approval success.
Creating compliant design history files demands multidisciplinary expertise spanning biomechanical engineering, materials science, clinical medicine, and regulatory affairs. Orthopedic OEM partners with mature design capabilities maintain template structures that ensure all required elements are addressed while providing flexibility for device-specific considerations. Risk management documentation integrating ISO 14971 methodologies forms a critical component, systematically identifying potential hazards, evaluating their severity and probability, and documenting risk mitigation measures implemented in the final design. This structured approach to safety assessment satisfies regulatory expectations while genuinely improving device safety profiles.
Post-Market Surveillance Obligations That Extend Beyond Sale
Regulatory compliance in orthopedic OEM manufacturing extends throughout the product lifecycle, including post-market surveillance activities that monitor device performance after commercial distribution. Medical device regulations worldwide require manufacturers to establish complaint handling systems that capture, investigate, and trend adverse events and product quality issues. For orthopedic OEM providers, this obligation means implementing mechanisms to receive information from distribution partners and end users, conducting root cause analyses when problems arise, and reporting serious incidents to regulatory authorities within specified timeframes.
Effective post-market surveillance generates valuable intelligence that drives continuous improvement in orthopedic OEM manufacturing processes. Trending analysis of complaint data may reveal emerging patterns indicating process drift, design vulnerabilities, or user training needs before they escalate into serious safety issues. This feedback loop enables proactive interventions including corrective and preventive actions that enhance product reliability and customer satisfaction. Leading orthopedic OEM manufacturers view post-market surveillance not as regulatory burden but as strategic competitive advantage, providing insights that inform next-generation product development and manufacturing optimization initiatives.
The Business Case for Uncompromising Standards in Orthopedic OEM
Risk Mitigation Through Quality Excellence
The financial implications of material, process, or compliance failures in orthopedic OEM manufacturing far exceed the incremental costs of implementing rigorous standards. Product recalls in the medical device industry carry catastrophic consequences including direct costs for retrieval and replacement, legal liability for patient injuries, regulatory sanctions that may restrict market access, and reputational damage that erodes customer confidence and future business opportunities. For orthopedic devices implanted in patients, recall execution becomes particularly complex and may require surgical revision procedures, amplifying costs and patient harm.
Investing in material verification programs, advanced process controls, and compliance infrastructure represents risk mitigation that protects both orthopedic OEM manufacturers and their customers from these scenarios. The cost differential between standard and medical-grade materials, between conventional and precision machining capabilities, and between basic and comprehensive quality systems is marginal compared to potential recall exposure. Experienced orthopedic device companies recognize this economic reality and prioritize manufacturing partners who demonstrate unwavering commitment to quality standards, even when this commitment requires higher pricing than less scrupulous competitors.
Market Differentiation Through Regulatory Readiness
Orthopedic OEM manufacturers with robust compliance infrastructure provide strategic value beyond manufacturing execution, serving as regulatory knowledge resources that accelerate product development and market entry timelines. Their familiarity with global regulatory requirements enables early identification of potential approval obstacles, allowing design modifications during development rather than after costly premarket submission rejections. This regulatory readiness becomes particularly valuable for emerging orthopedic device companies lacking internal regulatory expertise, where partnership with an experienced orthopedic OEM provider effectively extends their capabilities without requiring permanent staff expansion.
The competitive advantage extends to supporting multi-market regulatory strategies where devices require approval in multiple jurisdictions with varying requirements. Orthopedic OEM partners familiar with FDA, European MDR, and other regulatory frameworks can guide design decisions that maximize global compatibility, avoiding region-specific variants that increase inventory complexity and manufacturing costs. This strategic perspective transforms the orthopedic OEM relationship from transactional supplier arrangement to collaborative partnership that influences product success at fundamental levels.
Long-Term Partnership Value Through Operational Consistency
Material, process, and compliance excellence in orthopedic OEM manufacturing creates operational stability that enables long-term business planning and partnership development. When manufacturing partners consistently deliver on-specification products meeting agreed timelines, medical device companies can confidently commit to market launch dates, inventory planning parameters, and capacity expansion investments. This predictability reduces safety stock requirements, minimizes expediting costs associated with supply disruptions, and enables efficient working capital management.
Conversely, orthopedic OEM providers demonstrating inconsistent quality or compliance lapses introduce uncertainty that forces customers to maintain expensive contingencies or develop alternative sources, diluting the business relationship and eroding mutual investment incentives. The trust established through years of flawless execution cannot be easily replicated, creating switching costs that benefit both parties. Medical device companies gain supply security and manufacturing expertise, while orthopedic OEM manufacturers enjoy stable order volumes supporting workforce retention and capital equipment investments that further enhance capabilities.
FAQ
Why is biocompatibility testing mandatory for all orthopedic OEM materials even when using common alloys?
Biocompatibility testing remains mandatory regardless of material familiarity because slight variations in alloy composition, processing history, or surface treatments can significantly alter biological response. Even materials with established safety profiles require testing to verify that specific production lots and finishing processes do not introduce contaminants or surface characteristics that trigger adverse reactions. Regulatory authorities require this testing to ensure patient safety is based on actual device characteristics rather than assumptions about material class performance. Additionally, biocompatibility requirements vary by anatomical contact location and duration, making device-specific testing essential for appropriate risk assessment.
How do orthopedic OEM manufacturers maintain process control when scaling production volumes?
Maintaining process control during production scaling requires systematic validation of manufacturing processes at intended commercial volumes before full-scale implementation. Orthopedic OEM manufacturers conduct process validation studies demonstrating that critical quality attributes remain consistent across minimum, typical, and maximum production rates. This includes verifying that increased throughput does not compromise dimensional accuracy, that operator workload remains manageable without quality shortcuts, and that inspection resources scale proportionally with output. Statistical process control systems automatically flag deviations from established parameters regardless of production volume, ensuring early detection of any scale-related quality drift before significant quantities of non-conforming product are produced.
What specific compliance documentation should medical device companies request from orthopedic OEM partners?
Medical device companies should request ISO 13485 certification demonstrating quality management system compliance, material certificates of conformance verifying alloy specifications and biocompatibility testing, process validation reports proving manufacturing consistency, calibration records for measurement equipment used in production and inspection, and training records confirming operator competency for critical operations. Additionally, requesting copies of recent regulatory inspection reports and corrective action responses provides insight into compliance culture and issue resolution effectiveness. For custom development projects, design history file templates and risk management procedures indicate capability to support regulatory submissions. These documents collectively demonstrate an orthopedic OEM partner's technical capability and regulatory maturity.
Can orthopedic OEM manufacturers substitute materials to reduce costs without compromising device performance?
Material substitutions in orthopedic OEM manufacturing require comprehensive evaluation and formal change control procedures, as even seemingly minor variations can affect device safety and effectiveness. While cost reduction opportunities may exist through alternative material grades or suppliers, any substitution demands biocompatibility testing, mechanical property verification, and assessment of potential impacts on existing design validation studies. Regulatory requirements typically mandate that material changes undergo formal review and may require supplemental regulatory submissions depending on the extent of modification. Responsible orthopedic OEM manufacturers never implement material substitutions unilaterally but instead present proposals with supporting data allowing informed decisions about acceptable cost-performance tradeoffs while maintaining regulatory compliance and patient safety.
Table of Contents
- The Critical Role of Material Selection in Orthopedic OEM Manufacturing
- Process Control as the Foundation of Manufacturing Reliability
- Regulatory Compliance as a Market Access Imperative
- The Business Case for Uncompromising Standards in Orthopedic OEM
-
FAQ
- Why is biocompatibility testing mandatory for all orthopedic OEM materials even when using common alloys?
- How do orthopedic OEM manufacturers maintain process control when scaling production volumes?
- What specific compliance documentation should medical device companies request from orthopedic OEM partners?
- Can orthopedic OEM manufacturers substitute materials to reduce costs without compromising device performance?

