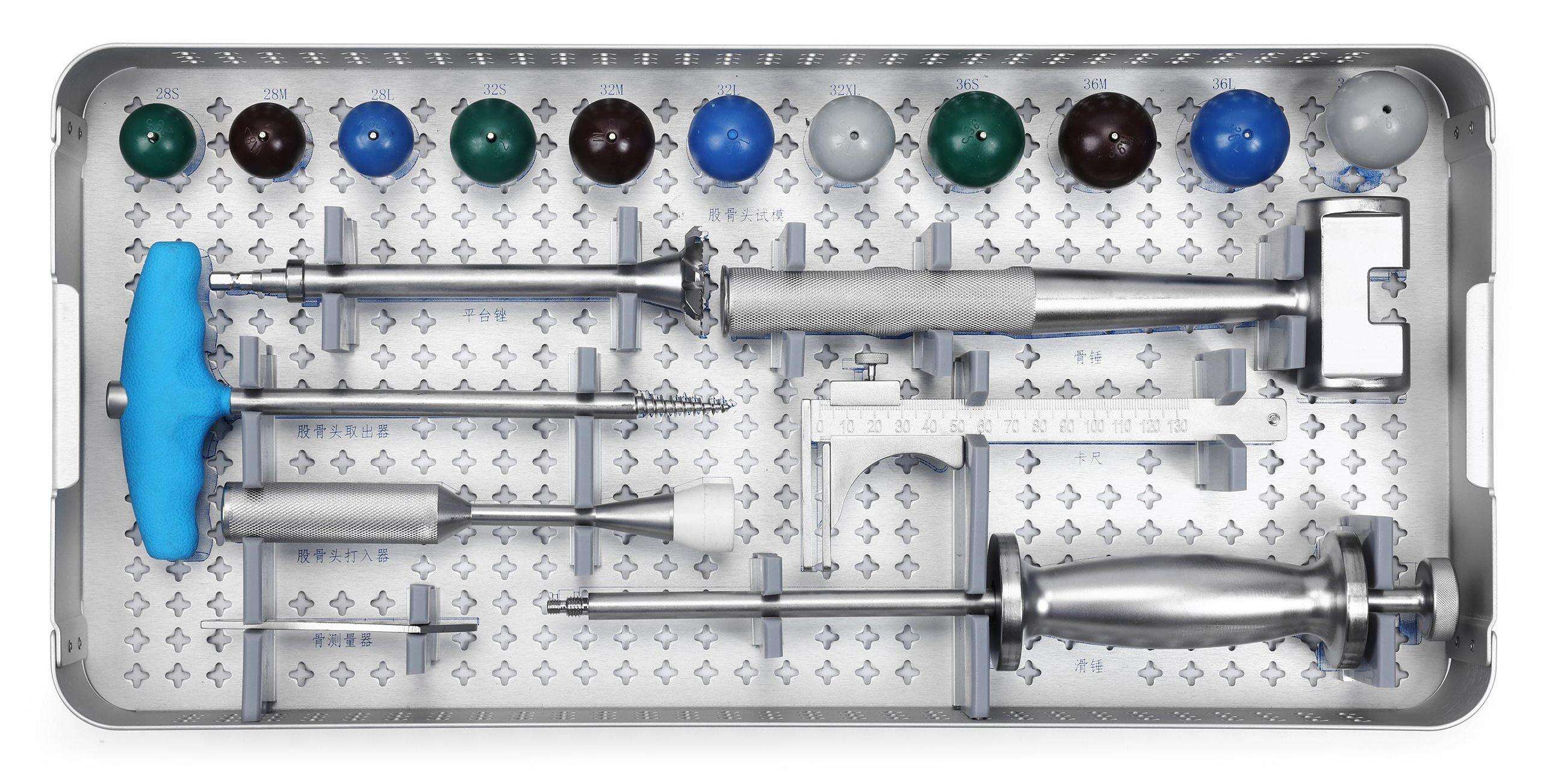
Posterior Pedicle Screw Fixation System Instrument Set
Precision. Stability. Customization for Global Spine Brands.
- Overview
- Recommended Products
Product Overview
The Posterior Pedicle Screw Fixation System Instrument Set is designed for posterior spinal fixation and fusion procedures across degenerative, deformity, and trauma cases.
This comprehensive system includes pedicle screws, connecting rods, crosslinks, reduction instruments, and ergonomically designed tools that enable accurate screw placement and secure fixation.
At Taruk Medical Instruments, we specialize in OEM/ODM manufacturing of spine surgical instrument systems.
Our engineering team supports international orthopedic brands and distributors with fully customized instrument solutions that meet both functional and regulatory requirements.
Key Technical Features
● Precision Machining and Tolerance Control – Each component is CNC-machined under strict ISO 13485 quality systems, maintaining dimensional accuracy within ±0.01 mm for reliable assembly and mechanical performance.
● Optimized Ergonomics – Surgeon-friendly handle designs and balanced weight distribution reduce intraoperative fatigue and enhance control.
● System Compatibility – Instruments can be tailored to match the client’s proprietary pedicle screw systems and implant configurations.
● Durability and Sterilization-Ready – High-grade stainless steel and titanium alloys provide excellent corrosion resistance and repeated autoclave compatibility.
● Surface Finishing Options – Mirror polishing, passivation, titanium anodizing, or customized coatings are available per customer specification.
● Modular Tray Design – Instruments are arranged in a streamlined layout for surgical efficiency and easy sterilization.
OEM/ODM Customization Capabilities
As an experienced OEM/ODM spine instrument manufacturer in China, Taruk provides flexible collaboration models for global partners:
● Design Co-Development – We work directly with client R&D teams to refine geometry, ergonomics, and system interfaces.
● Prototype & Sampling – 3D design review and rapid prototyping shorten your project validation cycle.
● Material & Process Customization – Titanium, medical stainless steel, or hybrid materials available; precision forging, CNC milling, laser marking supported.
● Private Labeling & Packaging – Customized branding, sterile or non-sterile packaging, tray labeling, and IFU printing.
● Small-Batch or Mass Production – From pilot batches for clinical trials to long-term volume supply with consistent quality.
● Documentation & Compliance Support – Full traceability records and production documentation to assist with CE/FDA submissions.
Manufacturing & Quality Assurance
Taruk Medical Instruments operates a modern manufacturing facility equipped with multi-axis CNC centers, ultrasonic cleaning lines, and Class 100,000 cleanroom assembly areas.
Our production management follows ISO 13485 standards.
Every batch of instruments undergoes:
● Incoming material inspection and chemical composition analysis
● Dimensional verification using coordinate measuring machines (CMM)
● Passivation and ultrasonic cleaning
● Laser marking for full traceability
● Final packaging under controlled environments
We maintain continuous improvement through statistical process control (SPC) and 100% visual and mechanical inspection before shipment.
Our robust quality system ensures that each instrument set leaving our facility meets the strictest international standards for orthopedic device manufacturing.
Taruk Manufacturing Advantages
● 20+ years of precision machining experience in orthopedic instrumentation
● Complete in-house production chain from raw material cutting to final packaging
● Engineering collaboration with global spine implant brands
● Annual production capacity exceeding 50,000 instrument sets
● Compliance with global export markets – products supplied to Europe, North America, and Asia-Pacific
Taruk’s value lies not only in manufacturing capability but also in technical communication and project management expertise — key factors that global medical brands expect from their strategic OEM partners in China.
Why Partner with Taruk
● Proven OEM/ODM supplier for orthopedic surgical instruments
● Transparent project workflow and documentation control
● Fast response to engineering revisions or urgent sampling requests
● Integrated packaging, sterilization, and logistics solutions
● Dedicated multilingual technical support and after-sales team
We help your brand accelerate product development, reduce manufacturing risks, and secure a reliable supply chain for your spine fixation systems.
Inquiry & Collaboration
Looking for a trusted manufacturing partner for your Posterior Pedicle Screw Fixation System Instrument Set?
Upload your design drawings or technical requirements, and our engineering team will provide a customized solution aligned with your brand’s implant system.
Contact Taruk Medical Instruments today to start your OEM/ODM project and experience reliable spine instrument manufacturing excellence.