The orthopedic implant industry stands at a pivotal transformation point where traditional contract manufacturing models no longer suffice to meet the complex demands of modern medical device innovation. As regulatory requirements tighten, patient expectations rise, and surgical techniques advance, orthopedic original equipment manufacturers (OEMs) increasingly seek manufacturing partners capable of delivering far more than production capacity. This shift from transactional supplier relationships to strategic value co-creation partnerships defines the new competitive landscape, with precision manufacturing emerging as the essential capability enabling this evolution. The ability to consistently produce orthopedic components within tolerances measured in microns, while simultaneously contributing design insights, material science expertise, and regulatory intelligence, separates commodity producers from transformative manufacturing partners.
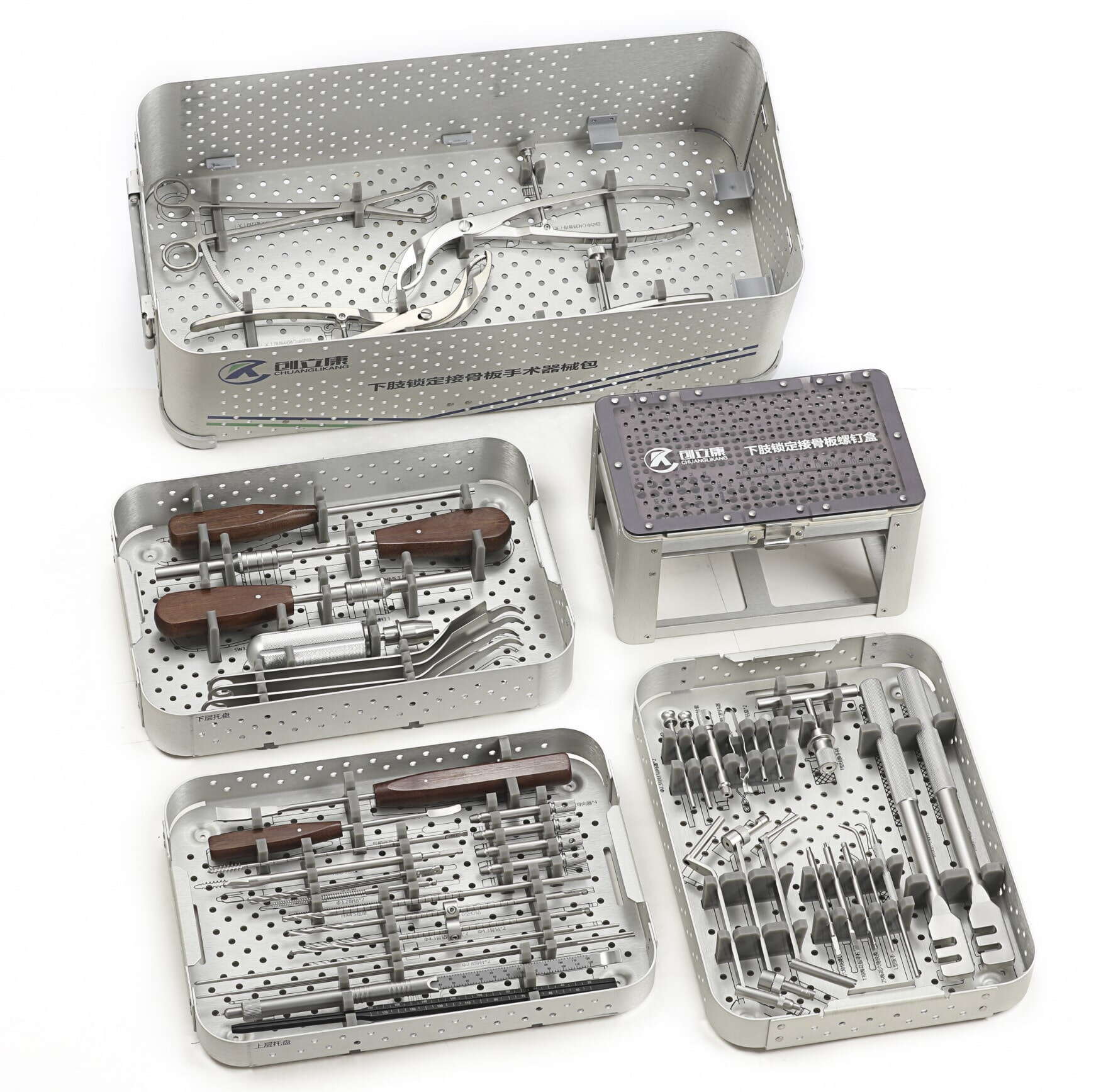
This fundamental restructuring of orthopedic manufacturing relationships reflects deeper industry dynamics that extend beyond operational efficiency. Precision manufacturing capabilities now serve as the foundation for collaborative innovation ecosystems where OEMs and manufacturing partners jointly develop next-generation implant systems, optimize surgical instrument designs, and accelerate time-to-market for breakthrough orthopedic solutions. Understanding how precision manufacturing enables this value co-creation model reveals why forward-thinking orthopedic companies are fundamentally rethinking their supplier selection criteria, partnership structures, and long-term strategic alliances. The manufacturing partner selection process has evolved from cost-per-unit comparisons to comprehensive evaluations of technical capability, innovation capacity, quality system maturity, and collaborative potential.
The Economic and Competitive Drivers Behind Manufacturing Partnership Evolution
Rising Development Costs and Market Access Barriers
Developing a new orthopedic implant system from concept through regulatory approval now routinely exceeds tens of millions of dollars in investment, with timelines stretching five to seven years for complex joint replacement systems. This resource-intensive development process creates powerful incentives for orthopedic OEMs to leverage external precision manufacturing expertise rather than building redundant in-house capabilities. Manufacturing partners with established quality systems, validated processes, and regulatory track records enable OEMs to redirect capital toward clinical research, surgeon education, and market development activities that directly differentiate their product portfolios. The financial calculus increasingly favors strategic manufacturing partnerships that compress development timelines and reduce technical risk.
Global market access requirements compound these development challenges as orthopedic companies navigate divergent regulatory frameworks across major markets. Precision manufacturing partners maintaining certifications across multiple jurisdictions—including FDA registration, ISO 13485 compliance, and CE marking capabilities—provide OEMs with turnkey pathways to international commercialization. This regulatory infrastructure represents substantial fixed investment that individual OEMs find difficult to justify for limited product portfolios, creating natural partnership opportunities where manufacturing specialists amortize compliance costs across multiple client relationships. The value proposition extends beyond cost sharing to include accumulated regulatory intelligence, established quality management systems, and proven audit histories that accelerate approval processes.
Technology Complexity and Specialized Manufacturing Knowledge
Modern orthopedic implants incorporate increasingly sophisticated material combinations, surface treatments, and geometric complexities that challenge conventional manufacturing approaches. Porous titanium structures promoting bone ingrowth, cobalt-chromium alloy components requiring precise metallurgical control, and polyethylene inserts with specific molecular weight distributions all demand specialized precision manufacturing knowledge rarely consolidated within single organizations. Manufacturing partners investing continuously in process development, equipment capabilities, and technical expertise create knowledge repositories that individual OEMs cannot economically replicate. These technical competencies transform manufacturing relationships from simple production transactions into knowledge-sharing collaborations where both parties contribute complementary expertise toward superior product outcomes.
The precision manufacturing requirements for orthopedic applications extend well beyond dimensional accuracy to encompass surface finish specifications, residual stress management, contamination control, and metallurgical consistency. Achieving these multidimensional quality attributes requires integrated process control spanning raw material qualification, machining parameter optimization, cleaning validation, and comprehensive inspection protocols. Manufacturing partners who have developed this systems-level expertise through years of focused orthopedic work offer OEMs access to mature capabilities without the extended learning curves and inevitable early failures that accompany internal capability development. The collaborative value emerges when manufacturing partners proactively suggest design modifications that improve manufacturability, enhance performance characteristics, or reduce production costs while maintaining clinical efficacy.
Market Responsiveness and Portfolio Flexibility
Orthopedic market dynamics increasingly reward companies capable of rapid product iteration, customized solutions for specific surgeon preferences, and agile responses to competitive innovations. Traditional vertically integrated manufacturing models, with their substantial fixed asset investments and organizational inertia, struggle to deliver this market responsiveness. Precision manufacturing partnerships enable OEMs to maintain lean, flexible product development organizations that can quickly prototype new designs, test alternative materials, and scale production in alignment with actual market demand rather than capacity planning forecasts. This operational flexibility becomes particularly valuable in specialty orthopedic segments where product lifecycles have shortened and surgeon preference patterns shift rapidly.
The portfolio management advantages extend beyond individual product development to encompass strategic resource allocation across entire implant system families. OEMs partnering with precision manufacturing specialists can simultaneously advance multiple development programs without the capital constraints and capacity limitations inherent in dedicated internal manufacturing facilities. This parallel development capability accelerates overall innovation velocity, enabling companies to maintain competitive product pipelines across multiple anatomical segments, surgical approaches, and patient populations. The manufacturing partnership model effectively converts fixed manufacturing costs into variable expenses that scale directly with commercial success, reducing financial risk during product launches and market development phases.
Precision Manufacturing as the Technical Foundation for Value Co-Creation
Dimensional Accuracy and Geometric Complexity Management
At the core of orthopedic precision manufacturing lies the fundamental capability to consistently produce components meeting extraordinarily tight dimensional tolerances across complex three-dimensional geometries. Joint replacement components often require tolerances within 0.01mm to ensure proper articulation, load distribution, and long-term implant survival. Achieving this precision manufacturing standard demands advanced machining centers with thermal stability, sophisticated fixturing systems, and metrology capabilities that can verify geometric accuracy in multiple dimensions simultaneously. Manufacturing partners investing in this equipment infrastructure provide OEMs access to capabilities that would require multimillion-dollar capital commitments if developed internally.
The geometric complexity of modern orthopedic implants frequently incorporates anatomically contoured surfaces, porous structures with controlled porosity gradients, and intricate features designed to optimize biological fixation or soft tissue integration. Producing these complex geometries reliably requires precision manufacturing expertise spanning multiple process technologies including five-axis machining, additive manufacturing, casting, and forging. Manufacturing partners who have developed integrated process chains combining these technologies offer OEMs design freedom to optimize implant performance without manufacturing constraints limiting innovation. This technical capability transforms the design process from a manufacturing-constrained exercise into a clinically optimized development program where form follows function rather than production limitations.
Material Science Application and Process Validation
Orthopedic implant performance depends critically on precise material properties including grain structure, phase composition, residual stress states, and surface chemistry. Precision manufacturing partners specializing in orthopedic applications develop deep expertise in material processing parameters that influence these properties. Understanding how thermal cycles during machining affect titanium microstructure, how forging parameters influence cobalt-chromium grain size, or how surface preparation impacts osseointegration represents specialized knowledge accumulated through extensive process development and validation work. OEMs partnering with manufacturing specialists access this material science expertise without building dedicated metallurgical laboratories and process development teams.
Process validation requirements for orthopedic implants demand rigorous documentation demonstrating that manufacturing processes consistently produce components meeting all specifications. Precision manufacturing partners maintaining validated processes across multiple implant categories provide OEMs with established quality frameworks that dramatically accelerate new product introductions. The validation infrastructure includes installation qualification, operational qualification, and performance qualification protocols for all manufacturing equipment, along with process capability studies demonstrating statistical control. This quality system maturity enables collaborative development approaches where manufacturing partners contribute validation expertise during design phases, identifying potential manufacturing challenges before they impact project timelines or require costly design iterations.
Advanced Inspection and Quality Verification Systems
Verifying that manufactured orthopedic components meet design specifications requires measurement systems often more sophisticated than the production equipment itself. Coordinate measuring machines with sub-micron accuracy, computed tomography scanners capable of internal geometry verification, and surface profilometers measuring roughness at nanometer scales represent the metrology infrastructure supporting precision manufacturing operations. Manufacturing partners investing in comprehensive inspection capabilities provide OEMs with objective evidence that components meet specifications while simultaneously generating data streams that enable continuous process improvement. The measurement data generated during routine production creates opportunities for statistical analysis identifying process trends, predicting maintenance requirements, and optimizing manufacturing parameters.
Beyond dimensional verification, orthopedic precision manufacturing requires material verification, surface treatment validation, and cleanliness confirmation. Techniques including X-ray fluorescence spectroscopy, scanning electron microscopy, and residue extraction testing ensure that manufactured components meet all material and surface quality requirements. Manufacturing partners maintaining these analytical capabilities enable OEMs to verify product conformance comprehensively without maintaining duplicate testing laboratories. The collaborative value emerges when manufacturing partners proactively apply these analytical tools during process development, identifying root causes of quality variations and implementing corrective actions that improve overall process capability.
Collaborative Innovation Models Enabled by Manufacturing Partnerships
Design for Manufacturability Integration
Traditional sequential product development models where design teams complete component specifications before engaging manufacturing specialists inevitably result in costly design iterations, manufacturing challenges, and compromised performance characteristics. Leading orthopedic OEMs now embrace concurrent engineering approaches where precision manufacturing partners participate in design discussions from initial concept stages. This early involvement enables manufacturing specialists to suggest design modifications that improve production efficiency, enhance quality consistency, or enable use of superior materials while maintaining clinical performance objectives. The collaborative design process transforms manufacturing partners from passive recipients of specifications into active contributors shaping product architectures.
Design for manufacturability contributions from precision manufacturing partners often identify opportunities for part consolidation, tolerance relaxation in non-critical features, or alternative manufacturing processes that reduce costs without compromising functionality. A manufacturing partner with extensive experience producing precision manufacturing solutions might suggest design features that facilitate automated inspection, reduce machining cycle times, or improve material utilization rates. These seemingly minor modifications can generate substantial cost savings when multiplied across thousands of implants while simultaneously improving manufacturing quality through simplified processes with fewer opportunities for error. The cumulative impact of manufacturing-informed design optimization often differentiates commercially successful products from technically equivalent designs that struggle with manufacturing challenges.
Material Development and Alternative Process Exploration
The orthopedic industry continually seeks improved materials offering enhanced biocompatibility, superior mechanical properties, or better imaging compatibility. Developing manufacturing processes for novel materials represents substantial technical risk that many OEMs hesitate to undertake independently. Precision manufacturing partners can amortize material development investments across multiple client relationships, creating economic models that support exploratory process development. Collaborative material development programs leverage manufacturing partner process expertise and OEM clinical knowledge to evaluate new material options, develop manufacturing processes, and validate performance characteristics. These joint development efforts accelerate material innovation while distributing technical and financial risks across partnership structures.
Beyond novel materials, manufacturing partnerships enable exploration of alternative production processes that may offer performance or cost advantages. Additive manufacturing technologies, for example, enable orthopedic implant geometries impossible to produce through conventional machining while potentially reducing material waste and lead times. Precision manufacturing partners investing in emerging technologies provide OEMs with low-risk opportunities to evaluate these processes through prototype development and limited production runs before committing to full-scale implementation. The partnership model allows OEMs to maintain technology neutrality, selecting optimal manufacturing approaches for specific applications rather than justifying capital investments in particular process technologies.
Regulatory Strategy and Submission Support
Regulatory submissions for orthopedic devices require extensive manufacturing documentation demonstrating process controls, quality systems, and production consistency. Precision manufacturing partners experienced in regulatory submissions contribute substantial value by providing comprehensive manufacturing sections for regulatory filings, supporting regulatory agency inspections, and maintaining documentation systems that facilitate efficient submission preparation. This regulatory expertise proves particularly valuable for smaller orthopedic companies with limited regulatory affairs resources or for companies entering new geographic markets with unfamiliar regulatory frameworks. The collaborative approach to regulatory strategy ensures that manufacturing considerations are appropriately addressed in regulatory submissions while manufacturing partners remain accountable for maintaining compliant production systems.
Manufacturing partner regulatory contributions extend beyond documentation to include proactive quality system management that prevents compliance issues. Established precision manufacturing partners maintain mature quality management systems with comprehensive corrective and preventive action protocols, supplier qualification programs, and change control procedures that meet or exceed regulatory expectations. OEMs benefit from these quality systems without bearing the full organizational overhead of maintaining equivalent internal capabilities. The partnership structure creates appropriate accountability where manufacturing partners bear responsibility for production quality while OEMs maintain overall product responsibility and market authorization. This division of responsibilities enables efficient organizational structures optimized around core competencies rather than comprehensive vertical integration.
Operational Excellence and Continuous Improvement in Manufacturing Partnerships
Supply Chain Integration and Transparency
Effective value co-creation requires supply chain visibility extending beyond the immediate manufacturing partner to encompass raw material suppliers, surface treatment providers, and sterilization service providers. Leading precision manufacturing partners develop integrated supply networks with qualified suppliers for critical materials and processes, creating turnkey solutions that simplify OEM supply chain management. This supply chain coordination reduces procurement complexity, improves material traceability, and ensures consistent raw material quality. The manufacturing partner assumes responsibility for supplier qualification, incoming inspection, and material inventory management, allowing OEMs to focus on product commercialization rather than supply chain logistics.
Digital integration increasingly enables real-time supply chain transparency where OEMs access production status, quality data, and inventory information through shared information systems. This visibility supports collaborative production planning, enables proactive issue resolution, and provides objective data for continuous improvement initiatives. Precision manufacturing partners investing in digital infrastructure create information-sharing capabilities that transform opaque supply relationships into transparent collaborative partnerships. The data generated through integrated systems supports analytical approaches to process optimization, quality improvement, and capacity planning that benefit both manufacturing partners and OEM clients through improved operational efficiency.
Statistical Process Control and Predictive Quality Management
Traditional quality management approaches based on final inspection and reactive corrective actions are giving way to statistical process control methodologies that predict and prevent quality issues before they occur. Precision manufacturing partners implementing advanced statistical methods continuously monitor process parameters, identify trends indicating potential quality drift, and intervene proactively to maintain process stability. This predictive quality management approach reduces scrap rates, improves process capability, and generates confidence in manufacturing consistency. OEMs partnering with statistically sophisticated manufacturers benefit from superior quality performance without maintaining internal statistical expertise or process monitoring infrastructure.
The data streams generated through statistical process control create opportunities for machine learning applications that further enhance quality prediction and process optimization. Precision manufacturing partners at the forefront of digital manufacturing apply advanced analytics to historical process data, identifying subtle correlations between process parameters and quality outcomes that enable continuous process refinement. These analytical capabilities represent substantial technical investments that manufacturing specialists can justify through application across multiple product lines and client relationships. OEMs benefit from these advanced capabilities without bearing full development costs, accessing manufacturing intelligence that would be economically impractical to develop for limited product portfolios.
Scalability Planning and Capacity Management
Orthopedic product launches face inherent demand uncertainty where initial market uptake may significantly exceed or fall short of planning forecasts. Precision manufacturing partnerships provide demand flexibility that mitigates this commercial risk by enabling production scaling aligned with actual market demand rather than fixed capacity commitments. Manufacturing partners serving multiple clients can balance capacity utilization across product portfolios, absorbing demand variability that would create operational challenges for dedicated internal manufacturing facilities. This capacity flexibility proves particularly valuable during product launches where demand patterns remain uncertain or for seasonal products with predictable utilization cycles.
Long-term capacity planning in manufacturing partnerships requires collaborative forecasting processes where OEMs share market intelligence and growth projections while manufacturing partners commit to capability development and capacity expansion. These planning dialogues create mutual commitments that balance OEM needs for assured supply with manufacturing partner requirements for capacity utilization visibility. Sophisticated manufacturing partnerships incorporate contractual mechanisms addressing capacity reservation, volume commitments, and flexibility provisions that align incentives and distribute risks appropriately. The partnership maturity required for effective capacity planning develops over time as trust builds and both parties demonstrate commitment to long-term collaboration rather than opportunistic short-term optimization.
Strategic Partnership Management and Relationship Evolution
Intellectual Property Protection and Confidentiality Management
Collaborative innovation models raise important intellectual property considerations as design information, process knowledge, and technical insights flow between organizations. Effective precision manufacturing partnerships establish clear intellectual property frameworks that protect OEM product designs while recognizing manufacturing partner process innovations. Well-structured agreements delineate ownership of background intellectual property, define treatment of foreground intellectual property developed during collaboration, and establish confidentiality obligations protecting proprietary information. These frameworks enable open technical collaboration while preserving appropriate intellectual property protection for both parties.
Beyond contractual protections, operational practices including physical security measures, information system access controls, and employee confidentiality training ensure that proprietary information receives appropriate safeguarding. Precision manufacturing partners serving multiple orthopedic clients implement rigorous information barriers preventing inadvertent knowledge transfer between competing clients. These operational protections build confidence enabling OEMs to share sensitive design information and development plans necessary for effective collaboration. The maturity of information protection practices often distinguishes professional manufacturing partners from less sophisticated suppliers who may lack appropriate controls for managing confidential client information.
Performance Measurement and Continuous Improvement Frameworks
Successful long-term manufacturing partnerships require objective performance measurement systems that track quality, delivery, cost, and innovation contributions. Effective metrics encompass traditional manufacturing performance indicators including defect rates, on-time delivery percentages, and cost trends, while also capturing collaborative value creation through innovation metrics, design optimization contributions, and joint development program success. These multidimensional measurement frameworks provide balanced perspectives on partnership health, identifying improvement opportunities while recognizing positive contributions across operational and strategic dimensions.
Regular business reviews provide structured forums for discussing performance trends, addressing emerging challenges, and planning future collaboration. These meetings create opportunities for senior leadership engagement, demonstrating organizational commitment to partnership success while enabling strategic discussions that extend beyond day-to-day operational issues. Precision manufacturing partnerships that institutionalize regular strategic dialogue develop deeper relationships more resistant to short-term performance fluctuations or organizational changes. The relationship maturity developed through consistent engagement creates partnership resilience and long-term stability that benefits both organizations through reduced relationship management overhead and improved collaborative efficiency.
Partnership Evolution Toward Strategic Integration
Manufacturing relationships often evolve through maturity stages from transactional supplier interactions toward deeply integrated strategic partnerships. Initial engagements typically focus on specific component production with limited collaboration beyond specification compliance. As relationships mature and mutual trust develops, collaboration expands to include design input, process development, and joint problem-solving. The most advanced partnerships achieve strategic integration where manufacturing partners participate in product planning, contribute to technology roadmaps, and share in commercial success through risk-sharing commercial models. This partnership evolution creates substantial competitive advantages as the collaborative efficiency and mutual understanding developed over time become difficult for competitors to replicate.
Strategic manufacturing partnerships may incorporate joint investment in capability development, exclusive capacity commitments, or shared intellectual property arrangements that align long-term interests. These deep integration models work best when both organizations commit to long-term collaboration, maintain compatible corporate cultures, and demonstrate consistent reliability in meeting commitments. The organizational investment required for strategic integration makes partner selection critically important as these relationships prove difficult to change once established. OEMs pursuing strategic manufacturing partnerships must evaluate not only current technical capabilities but also cultural compatibility, strategic alignment, and organizational stability to ensure partnership longevity.
FAQ
What distinguishes precision manufacturing from standard contract manufacturing in orthopedic applications?
Precision manufacturing in orthopedic applications refers to specialized capabilities producing components with extremely tight tolerances, complex geometries, and rigorous quality requirements specific to medical implants. While standard contract manufacturing focuses primarily on production efficiency and cost optimization, precision manufacturing emphasizes dimensional accuracy within microns, comprehensive process validation, advanced material expertise, and regulatory compliance. Precision manufacturers invest substantially in metrology equipment, process control systems, and quality infrastructure that enable consistent production of components meeting orthopedic implant specifications. The value proposition extends beyond manufacturing execution to include design collaboration, material development support, and regulatory documentation contributions that characterize true value co-creation partnerships rather than simple production transactions.
How do orthopedic OEMs evaluate manufacturing partners for strategic collaboration potential?
Evaluating manufacturing partners for strategic collaboration requires assessment across multiple dimensions beyond traditional supplier qualification criteria. Technical capability evaluation examines machining equipment sophistication, metrology infrastructure, process validation maturity, and demonstrated experience with similar orthopedic applications. Quality system assessment reviews ISO 13485 certification, FDA registration status, audit history, and corrective action effectiveness. Innovation capacity evaluation considers engineering resources, material science expertise, and track record of collaborative development contributions. Cultural compatibility assessment examines communication practices, responsiveness patterns, and alignment of business values. Financial stability analysis ensures long-term viability and capacity for necessary capability investments. Leading OEMs employ multifunctional evaluation teams including engineering, quality, regulatory, and procurement representatives to assess these diverse factors comprehensively before establishing strategic manufacturing partnerships.
What commercial models support value co-creation in precision manufacturing partnerships?
Value co-creation partnerships often employ commercial models that extend beyond simple unit pricing to align incentives around innovation, quality, and long-term collaboration. Traditional purchase order relationships may evolve toward longer-term supply agreements providing volume commitments that enable manufacturing partners to justify capability investments. Cost-sharing arrangements for development programs distribute financial risk while creating shared ownership of resulting intellectual property. Gain-sharing models reward manufacturing partners for cost reduction ideas or process improvements that benefit OEM profitability. Performance-based pricing incorporates quality metrics, delivery performance, or innovation contributions into compensation formulas. Some advanced partnerships establish joint venture structures for specific product lines, creating direct alignment of commercial interests. The optimal commercial model depends on product characteristics, development stage, competitive dynamics, and relationship maturity, often evolving as partnerships deepen over time.
How do precision manufacturing partnerships address the challenge of maintaining multiple OEM client relationships without creating conflicts of interest?
Professional precision manufacturing partners implement comprehensive conflict management systems to serve multiple orthopedic OEM clients ethically and effectively. Information barriers including physical separation of manufacturing areas, restricted access controls, and dedicated project teams prevent inadvertent knowledge transfer between competing clients. Confidentiality agreements with clear intellectual property provisions protect proprietary designs and development information. Client acceptance processes evaluate potential conflicts before accepting new business, declining engagements that would create unmanageable competitive tensions. Transparent communication about client base composition, though without revealing specific client identities, enables OEMs to assess potential concerns. Many manufacturing partners develop complementary client portfolios serving different anatomical segments or surgical approaches that minimize direct competitive overlap. The ability to manage multiple client relationships without conflicts demonstrates organizational maturity and professionalism that characterizes leading precision manufacturing partners in the orthopedic industry.
Table of Contents
- The Economic and Competitive Drivers Behind Manufacturing Partnership Evolution
- Precision Manufacturing as the Technical Foundation for Value Co-Creation
- Collaborative Innovation Models Enabled by Manufacturing Partnerships
- Operational Excellence and Continuous Improvement in Manufacturing Partnerships
- Strategic Partnership Management and Relationship Evolution
-
FAQ
- What distinguishes precision manufacturing from standard contract manufacturing in orthopedic applications?
- How do orthopedic OEMs evaluate manufacturing partners for strategic collaboration potential?
- What commercial models support value co-creation in precision manufacturing partnerships?
- How do precision manufacturing partnerships address the challenge of maintaining multiple OEM client relationships without creating conflicts of interest?

