The orthopedic device industry stands at a pivotal juncture where regulatory demands and clinical precision requirements converge with unprecedented intensity. Modern orthopedic device OEM manufacturers face a landscape transformed by stringent international quality standards, evolving biocompatibility protocols, and surgical precision expectations measured in micrometers rather than millimeters. This transformation has fundamentally altered how contract manufacturers approach product development, pushing them beyond traditional fabrication roles into strategic partnerships that balance regulatory compliance with technical excellence. Today's orthopedic device OEM providers must navigate complex certification pathways while delivering components that meet exacting tolerances for implants, instrumentation systems, and surgical tools that directly impact patient outcomes.
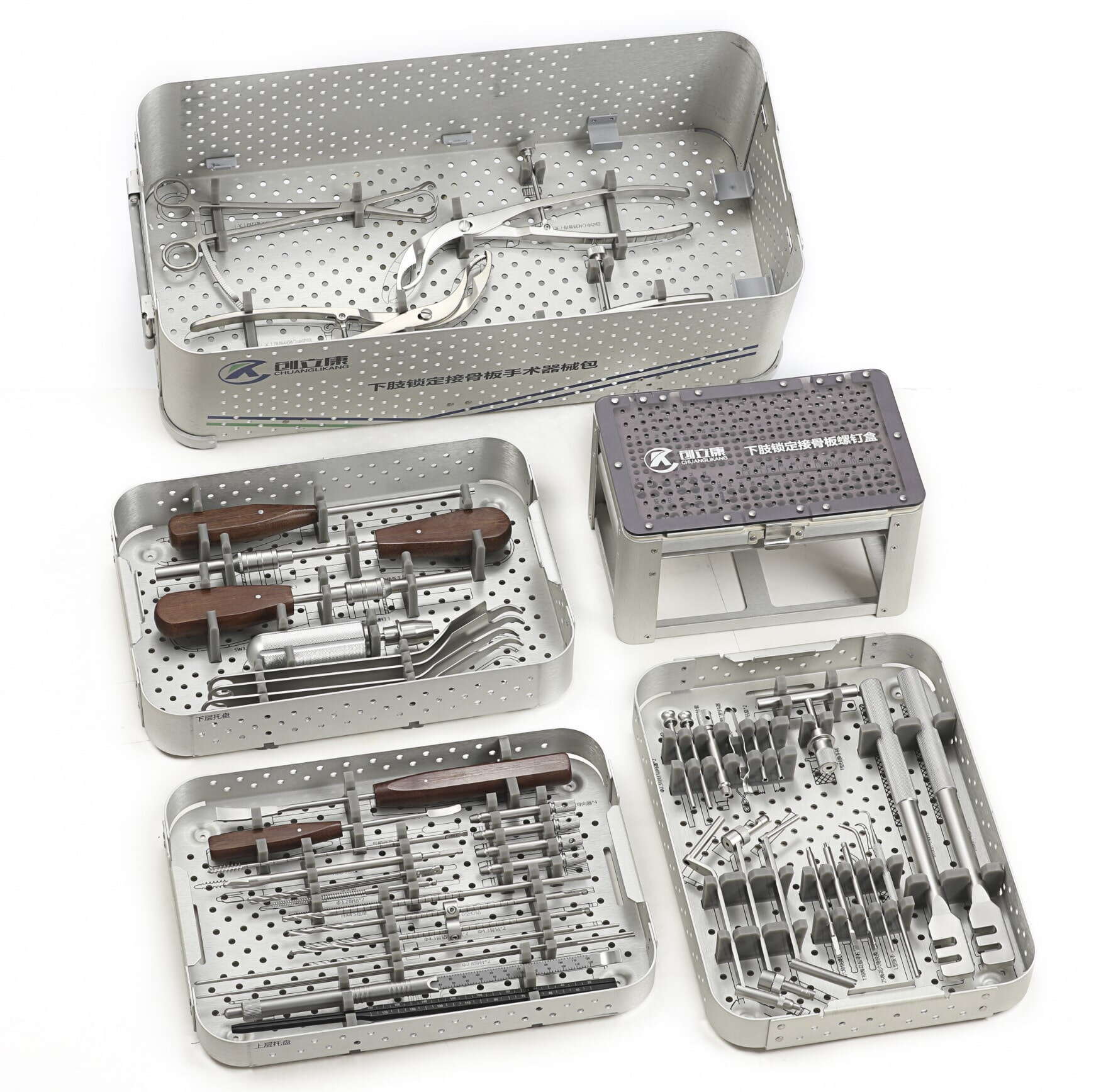
The dual imperatives of compliance and precision have redefined the competitive landscape for orthopedic device OEM operations, creating distinct barriers to entry and separating capable manufacturers from those merely offering machining services. Leading manufacturers now invest extensively in quality management systems that integrate real-time monitoring, traceability protocols, and validation frameworks extending from raw material procurement through final inspection. These operational enhancements reflect the reality that orthopedic devices—whether trauma plates, spinal implants, or surgical instrument sets—demand manufacturing approaches where regulatory documentation and dimensional accuracy receive equal strategic priority. Understanding how manufacturers successfully navigate these parallel requirements provides essential insight for medical device companies seeking reliable production partners capable of supporting both market access and clinical performance objectives.
Regulatory Compliance Architecture in Modern Orthopedic Manufacturing
Quality Management System Integration for Medical Device Standards
Contemporary orthopedic device OEM facilities operate under comprehensive quality management systems that extend far beyond basic ISO 13485 certification. These systems incorporate risk management protocols aligned with ISO 14971, biocompatibility testing frameworks conforming to ISO 10993 series standards, and product lifecycle documentation that satisfies FDA Quality System Regulation requirements and European Medical Device Regulation demands simultaneously. The integration depth separates competent manufacturers from basic contract shops, as true compliance requires embedded quality thinking throughout design transfer, process validation, and production execution rather than end-stage inspection approaches.
Leading manufacturers implement layered verification systems where incoming material inspections validate supplier certificates against actual testing, in-process controls monitor critical parameters at defined intervals, and final product audits employ statistical sampling methodologies that ensure batch consistency. This architecture creates documented evidence trails linking every production lot to specific material batches, operator qualifications, equipment calibration records, and environmental monitoring data. For orthopedic device OEM partners, this documentation infrastructure enables rapid response to regulatory inquiries, supports post-market surveillance requirements, and provides the traceability foundation necessary for global market distribution across multiple regulatory jurisdictions.
Certification Portfolio Management Across Global Markets
Successful orthopedic device OEM manufacturers maintain active certifications spanning multiple regulatory frameworks to support client market access strategies. This typically includes FDA registration with appropriate device class listings, CE marking under the Medical Device Regulation framework with notified body oversight, and increasingly Health Canada Medical Device Licensing and therapeutic goods administration registrations for Pacific markets. The certification portfolio extends beyond basic facility approvals to encompass product-specific technical files, clinical evaluation reports, and post-market surveillance protocols that satisfy differing regional requirements for orthopedic implants and instrumentation.
The strategic value of comprehensive certification coverage becomes apparent during new product introductions, where manufacturers with established regulatory pathways can compress time-to-market timelines substantially compared to facilities requiring new approvals. Experienced orthopedic device OEM partners leverage existing quality system infrastructure and regulatory relationships to streamline documentation preparation, coordinate notified body audits, and manage parallel submission processes across target markets. This regulatory agility particularly benefits medical device companies navigating the transition from development prototypes to commercial production, as established manufacturers provide regulatory roadmaps informed by previous product launches within similar device classifications.
Material Traceability and Biocompatibility Documentation
Material sourcing and verification represent critical compliance touchpoints where orthopedic device OEM manufacturers establish the foundation for regulatory submissions and post-market traceability. Leading facilities implement supplier qualification programs that extend beyond purchase agreements to include regular audits, material testing protocols, and certificate of conformance verification systems. For orthopedic applications involving titanium alloys, stainless steel grades, and cobalt-chromium materials, this means validating chemical composition, mechanical properties, and biocompatibility test results against international standards specifications before materials enter production workflows.
The biocompatibility documentation chain connects raw material certifications to finished device testing through batch-specific records that track material lots from ingot or bar stock through machining, surface treatment, and final sterilization processes. Modern orthopedic device OEM facilities maintain digital traceability systems that link component serial numbers to specific material certificates, enabling rapid investigation responses during post-market surveillance activities or adverse event reporting situations. This granular traceability approach satisfies regulatory requirements while providing manufacturers and their clients with risk management tools that support product liability defenses and continuous improvement initiatives based on field performance data analysis.
Precision Engineering Capabilities for Orthopedic Applications
Advanced Machining Technologies for Complex Geometries
Modern orthopedic device OEM manufacturing relies on multi-axis CNC machining centers capable of executing complex toolpaths with position accuracy measured in single-digit microns. Five-axis simultaneous machining enables production of anatomically contoured implant surfaces, undercut features in locking plate designs, and intricate instrument geometries that would be impossible or economically impractical using conventional three-axis approaches. These capabilities prove essential for current-generation orthopedic devices where biomechanical optimization drives design toward organic shapes and variable cross-sections that challenge traditional manufacturing methods.
The precision requirements extend beyond dimensional accuracy to encompass surface finish specifications that influence both biocompatibility and functional performance. Orthopedic implant surfaces contacting bone tissue require controlled roughness profiles that promote osseointegration, while articulating surfaces in joint replacement components demand mirror finishes minimizing wear particle generation. Leading orthopedic device OEM facilities employ specialized tooling strategies, cutting parameter optimization, and in-process measurement systems that verify surface characteristics during production rather than discovering nonconformance during final inspection. This process control approach reduces scrap rates while ensuring consistent surface quality across production batches.
Dimensional Verification and Inspection Protocols
Quality assurance in precision orthopedic manufacturing depends on inspection capabilities matching or exceeding production machine accuracies. Contemporary facilities deploy coordinate measuring machines with touch-probe and optical scanning capabilities providing three-dimensional verification against CAD models with resolution to two microns. This measurement technology enables complete dimensional characterization of complex implant geometries, verification of thread forms on bone screws, and validation of mating interfaces between orthopedic device OEM instrument components where functional assembly depends on precise tolerances.
Advanced inspection protocols incorporate statistical process control methodologies that monitor dimensional trends rather than simply sorting conforming from nonconforming parts. By tracking key characteristics across sequential production lots, manufacturers identify process drift before it generates out-of-specification components, enabling proactive machine adjustments that maintain process capability indices appropriate for medical device applications. For orthopedic device OEM partners, this statistical approach demonstrates process understanding and control maturity that regulatory auditors recognize as evidence of robust quality systems capable of consistent performance over extended production runs.
Material Processing and Heat Treatment Precision
Mechanical property achievement in orthopedic implants and instruments depends on precisely controlled heat treatment processes that transform material microstructures to specified strength, hardness, and toughness combinations. Solution annealing treatments for titanium components, precipitation hardening cycles for stainless steel instruments, and stress relief operations following machining all require furnace systems with temperature uniformity, atmosphere control, and thermal cycle documentation meeting stringent medical device manufacturing standards. Experienced orthopedic device OEM manufacturers maintain dedicated heat treatment facilities with regular calibration verification and process validation protocols demonstrating consistent property development across production batches.
The integration between machining operations and heat treatment processes presents scheduling and logistics challenges that capable manufacturers address through systematic workflow planning. Components requiring post-machining heat treatment must be planned with appropriate stock allowances for thermal distortion, while finish machining operations following heat treatment demand tool selection and cutting strategies appropriate for hardened materials. Leading facilities coordinate these process sequences using manufacturing execution systems that track individual component progress, trigger inspection holds at critical transitions, and maintain complete process documentation satisfying medical device quality system requirements for production record completeness.
Design Transfer Excellence and Engineering Collaboration
Prototype Development and Design Validation Support
The transition from conceptual designs to manufacturable orthopedic products represents a critical phase where experienced orthopedic device OEM partners provide substantial value beyond basic fabrication services. Design transfer protocols employed by sophisticated manufacturers include design for manufacturability reviews that identify potential production challenges, tolerance analysis verifying achievable dimensional control, and material selection guidance based on manufacturing process capabilities. This collaborative approach during development phases prevents costly redesign cycles during production scaling and establishes realistic expectations for achievable specifications given available manufacturing technologies.
Prototype production capabilities within advanced orthopedic device OEM facilities enable iterative design refinement using actual production materials and representative manufacturing processes rather than relying solely on rapid prototyping technologies with different material properties. This approach proves particularly valuable for orthopedic applications where mechanical performance testing requires components exhibiting authentic material behavior under physiological loading conditions. Manufacturers supporting comprehensive prototype services typically maintain flexible machining capacity separate from high-volume production lines, allowing rapid turnaround of design iterations without disrupting ongoing commercial manufacturing commitments.
Process Validation and Production Scaling Strategies
Regulatory compliance for orthopedic devices requires formal process validation demonstrating consistent production of components meeting all specified requirements. Competent orthopedic device OEM manufacturers approach validation systematically through installation qualification of equipment, operational qualification of processes, and performance qualification using production representative runs that generate statistical evidence of process capability. This validation framework establishes documented proof that manufacturing processes will consistently produce acceptable quality output when operated within defined parameters, satisfying fundamental regulatory requirements for medical device manufacturing.
The scaling transition from prototype quantities to commercial production volumes introduces challenges in maintaining dimensional consistency, surface quality, and material properties as batch sizes increase and production rhythms intensify. Experienced manufacturers address scaling through deliberate capacity planning that introduces production volume incrementally while monitoring key process indicators for stability. This measured approach identifies process adjustments necessary for volume production before committing to full commercial launch, reducing the risk of quality issues emerging during market introduction phases when product reputation and customer confidence are most vulnerable to manufacturing problems.
Engineering Documentation and Change Control Systems
Comprehensive technical documentation forms the communication backbone between medical device companies and their orthopedic device OEM manufacturing partners. Complete documentation packages include detailed engineering drawings with geometric dimensioning and tolerancing specifications, material callouts with applicable standards references, surface finish requirements with measurement methodologies, and inspection protocols defining acceptance criteria. Leading manufacturers implement structured document control systems ensuring production teams access current revision levels, obsolete versions are removed from circulation, and change histories are maintained for regulatory traceability and continuous improvement analysis.
Change control processes govern how design modifications, process improvements, and material substitutions are evaluated, approved, and implemented without disrupting regulatory compliance or quality system integrity. Formal change request procedures employed by sophisticated orthopedic device OEM facilities require impact assessment across multiple dimensions including regulatory implications, validation status effects, existing inventory disposition, and customer notification requirements. This systematic change management prevents unauthorized modifications while enabling continuous improvement initiatives that enhance product quality, reduce manufacturing costs, or improve delivery performance without compromising the controlled environment essential for medical device production.
Supply Chain Integration and Production Continuity
Strategic Supplier Relationships for Critical Materials
Reliable orthopedic device OEM manufacturing depends on stable supply chains for specialty materials including medical-grade titanium alloys, implant-quality stainless steels, and biocompatible surface treatment chemicals. Leading manufacturers develop strategic relationships with material suppliers that extend beyond transactional purchasing to include collaborative forecasting, capacity reservations, and quality partnership agreements ensuring material availability and consistency. These relationships prove particularly valuable during global supply disruptions when established supply chains and qualified supplier networks provide continuity that ad-hoc purchasing approaches cannot match.
Supplier qualification programs implemented by mature orthopedic device OEM facilities establish performance expectations, audit schedules, and corrective action protocols that maintain material quality and delivery reliability. Regular supplier assessments evaluate financial stability, quality system robustness, and technical capabilities ensuring the supply base can support both current production requirements and future growth trajectories. This proactive supplier management approach creates resilient supply chains capable of withstanding market volatility while maintaining the material traceability and documentation rigor that medical device regulatory frameworks demand.
Inventory Management and Production Scheduling Optimization
Efficient production operations in orthopedic device OEM manufacturing balance competing objectives of minimizing inventory carrying costs while maintaining material availability for responsive order fulfillment. Advanced manufacturers employ material requirements planning systems integrated with customer demand forecasts, production capacity models, and supplier lead time databases to optimize inventory positions across raw materials, work-in-process, and finished goods. This systematic approach reduces capital tied up in excess inventory while preventing stockouts that delay customer deliveries and disrupt surgical schedules depending on timely orthopedic device availability.
Production scheduling in facilities managing multiple customer programs requires sophisticated capacity allocation balancing routine replenishment orders against prototype development projects and new product introduction activities. Leading orthopedic device OEM partners employ scheduling algorithms that consider setup time optimization, batch size economics, and delivery date commitments to maximize equipment utilization while meeting customer service level agreements. This operational excellence translates to predictable lead times, reliable delivery performance, and transparent communication regarding production status that medical device companies require for their own planning processes and commitments to hospital customers.
Risk Mitigation Through Redundancy and Contingency Planning
Business continuity planning represents a critical yet often overlooked dimension of orthopedic device OEM capability assessment. Sophisticated manufacturers implement redundancy strategies including backup equipment for critical processes, alternative supplier qualifications for key materials, and cross-training programs ensuring workforce flexibility during personnel unavailability. These preparations prove invaluable during unexpected disruptions ranging from equipment failures and quality holds to natural disasters and pandemic-related workforce constraints that have tested supply chain resilience across global manufacturing sectors.
Contingency planning extends to data backup systems, quality record preservation, and disaster recovery protocols ensuring regulatory compliance documentation remains accessible even during facility disruptions. For medical device companies dependent on consistent orthopedic device OEM supply for their commercial operations, partner business continuity capabilities directly influence their own ability to meet customer commitments and maintain market presence during challenging circumstances. Due diligence evaluations of potential manufacturing partners increasingly include business continuity assessment as a critical selection criterion recognizing that technical capabilities matter little if production interruptions leave customers without reliable supply alternatives.
Technology Adoption and Future-Readiness Positioning
Additive Manufacturing Integration for Complex Geometries
Progressive orthopedic device OEM manufacturers are integrating metal additive manufacturing technologies alongside traditional subtractive machining to expand design freedom and enable patient-specific customization. Selective laser melting and electron beam melting systems produce titanium and cobalt-chromium components with internal lattice structures, organic surface textures, and geometric complexity unachievable through conventional manufacturing. While additive technologies currently serve specialized applications rather than replacing established machining processes, their growing maturity positions forward-thinking manufacturers to support next-generation orthopedic device designs incorporating biomimetic structures and personalized anatomical fitting.
The regulatory pathway for additively manufactured orthopedic devices presents unique challenges including process validation for powder bed fusion technologies, material property characterization for anisotropic build structures, and quality control methodologies for layer-by-layer construction. Experienced orthopedic device OEM facilities investing in additive capabilities develop specialized quality protocols addressing these technology-specific requirements while maintaining integration with existing quality management systems. This regulatory preparedness enables manufacturers to support clients exploring additive manufacturing opportunities without compromising compliance standards or introducing uncontrolled risks into established quality frameworks.
Digital Manufacturing and Industry 4.0 Implementation
Smart manufacturing technologies are transforming orthopedic device OEM operations through real-time process monitoring, predictive maintenance systems, and data analytics platforms that enhance quality control and operational efficiency. Connected machining centers stream production data enabling immediate detection of tool wear, thermal drift, and process parameter deviations before they generate nonconforming components. This proactive quality approach surpasses traditional statistical process control by identifying subtle process changes in real-time rather than through retrospective analysis of measured samples, fundamentally improving process control responsiveness.
Digital thread implementation creates seamless data connectivity from initial design files through production execution and final inspection records, establishing complete product genealogy documentation supporting regulatory traceability requirements. Advanced orthopedic device OEM manufacturers leverage these integrated systems to generate automated production documentation, streamline batch record compilation, and provide customers with real-time visibility into production status and quality metrics. This digital transformation enhances operational transparency while reducing administrative burden associated with manual documentation practices that historically consumed substantial resources in medical device manufacturing environments.
Continuous Improvement Culture and Innovation Partnership
Sustained excellence in orthopedic device OEM manufacturing requires organizational cultures embracing continuous improvement methodologies and collaborative innovation with customer partners. Leading facilities implement formal improvement programs combining lean manufacturing principles, Six Sigma statistical approaches, and employee suggestion systems that systematically identify and eliminate waste, reduce variation, and enhance process capability. These structured improvement initiatives generate cumulative gains in quality performance, cost efficiency, and delivery reliability that compound over multi-year customer relationships.
Innovation partnerships between medical device companies and their orthopedic device OEM manufacturers extend beyond transactional supplier relationships toward collaborative development models where manufacturing expertise informs product design decisions. Manufacturers contributing process knowledge during early design phases help optimize component geometries for producibility, suggest material alternatives offering cost or performance advantages, and identify manufacturing technologies enabling differentiated product features. This collaborative approach creates mutual value as device companies gain access to specialized manufacturing knowledge while manufacturers secure long-term business relationships with customers viewing them as strategic partners rather than interchangeable vendors.
FAQ
What certifications should orthopedic device OEM manufacturers maintain for global market access?
Comprehensive orthopedic device OEM manufacturers should maintain ISO 13485 certification as the foundation quality management system standard, supplemented by FDA registration for U.S. market access and CE marking under the Medical Device Regulation with notified body oversight for European markets. Additional certifications including Health Canada licensing, Japanese PMDA registration, and therapeutic goods administration approval expand market reach. Beyond facility-level certifications, manufacturers should maintain product-specific technical files and clinical evaluation documentation supporting device registrations across target regulatory jurisdictions.
How do precision requirements differ between orthopedic implants and surgical instruments?
Orthopedic implants typically demand tighter dimensional tolerances particularly for articulating surfaces, screw threads, and bone-contacting geometries where micrometer-level precision directly influences biomechanical performance and osseointegration. Surgical instruments generally allow slightly more generous tolerances except for critical functional features like driver engagement surfaces and locking mechanisms. However, instruments often require superior surface hardness and wear resistance since they endure repeated use and sterilization cycles, while most implants are single-use devices. Both product categories require equivalent documentation rigor and regulatory compliance despite these technical differences in precision specifications.
What lead times are typical for transitioning orthopedic device designs from development to commercial production?
The transition timeline from prototype development to validated commercial production typically spans six to twelve months for orthopedic devices with established material platforms and conventional manufacturing processes. This duration includes design finalization, tooling procurement, process validation execution, and initial production runs generating performance qualification data. More complex devices requiring new material qualifications, additive manufacturing processes, or extensive biocompatibility testing may extend timelines to eighteen months. Experienced orthopedic device OEM manufacturers with established quality systems and regulatory pathways can compress these timelines by leveraging existing infrastructure, though regulatory validation requirements impose minimum durations that cannot be arbitrarily accelerated regardless of manufacturing capability.
How do manufacturers ensure consistent quality across high-volume orthopedic device production runs?
Consistent quality in high-volume orthopedic device OEM manufacturing relies on validated processes operated within defined parameter windows, statistical process control monitoring detecting process drift, and layered inspection protocols verifying critical characteristics at multiple production stages. Manufacturers employ automated measurement systems for high-frequency dimensional verification, maintain rigorous equipment calibration schedules, and implement environmental controls for temperature and humidity affecting dimensional stability. Material traceability systems link every production batch to specific raw material lots, enabling rapid investigation of quality issues and supporting continuous improvement analysis identifying systematic variation sources requiring process adjustments.
Table of Contents
- Regulatory Compliance Architecture in Modern Orthopedic Manufacturing
- Precision Engineering Capabilities for Orthopedic Applications
- Design Transfer Excellence and Engineering Collaboration
- Supply Chain Integration and Production Continuity
- Technology Adoption and Future-Readiness Positioning
-
FAQ
- What certifications should orthopedic device OEM manufacturers maintain for global market access?
- How do precision requirements differ between orthopedic implants and surgical instruments?
- What lead times are typical for transitioning orthopedic device designs from development to commercial production?
- How do manufacturers ensure consistent quality across high-volume orthopedic device production runs?
