Orthopedic original equipment manufacturers face a pervasive challenge that quietly undermines product launches, regulatory timelines, and market confidence: precision anxiety. This condition emerges when OEMs partner with contract manufacturers who lack the capability, documentation rigor, or quality systems to consistently deliver implants and instruments within micron-level tolerances. For orthopedic devices where patient safety depends on exact dimensional accuracy, surface finish consistency, and material traceability, even minor deviations create cascading risks. Medical device contract manufacturing partnerships must address this anxiety systematically, transforming it from a source of sleepless nights into a competitive advantage through strategic vendor selection, process validation protocols, and collaborative quality frameworks.
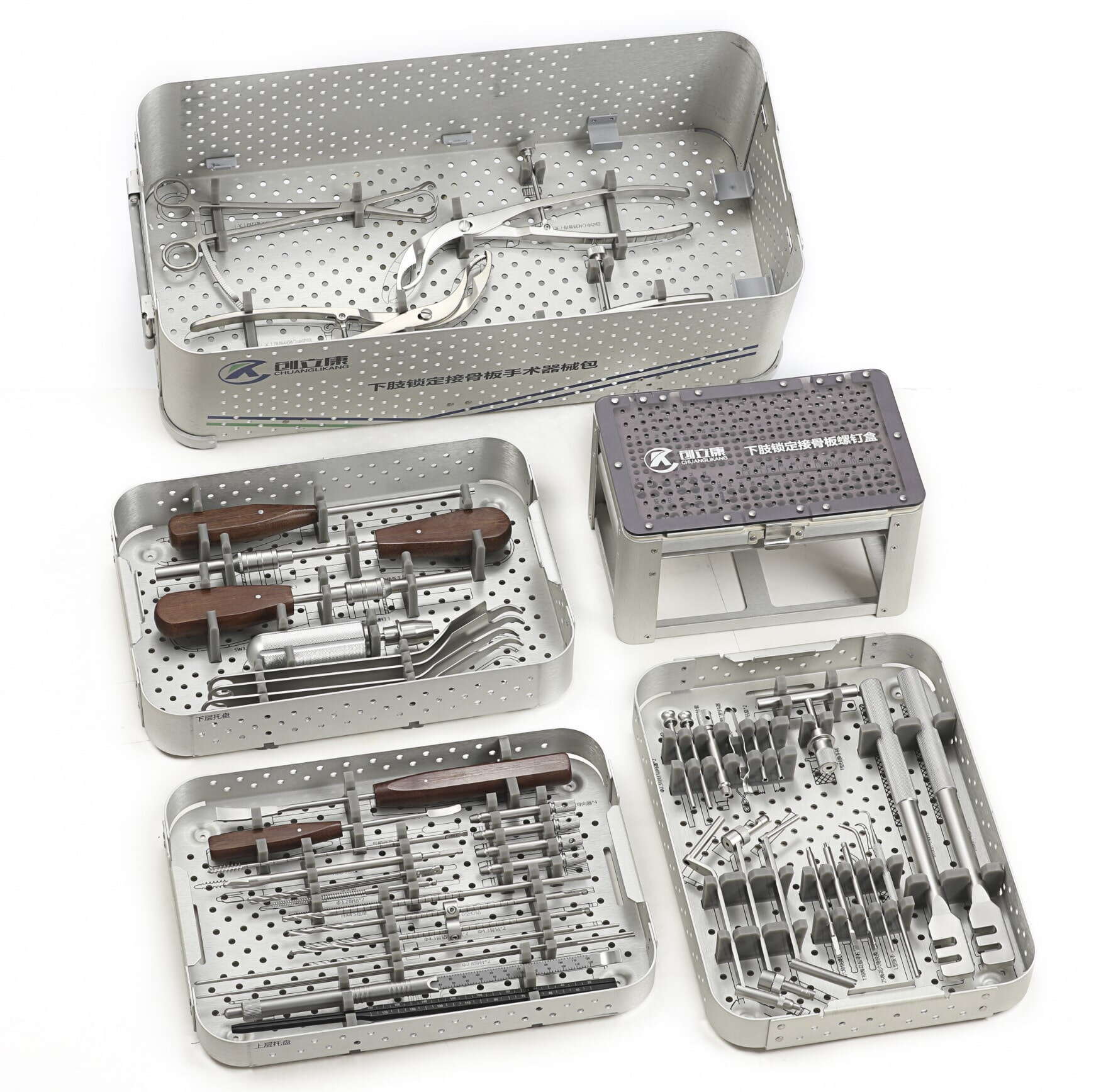
The stakes in orthopedic medical device contract manufacturing extend beyond commercial success to patient outcomes and regulatory compliance. When precision anxiety goes unaddressed, OEMs experience delayed FDA submissions, costly rework cycles, strained relationships with surgical staff, and erosion of brand reputation built over decades. This comprehensive guide examines the root causes of precision anxiety in contract manufacturing relationships, explores validation strategies that transform concern into confidence, and provides actionable frameworks for orthopedic OEMs to establish manufacturing partnerships that deliver consistent quality while maintaining regulatory compliance and cost efficiency.
Understanding the Root Causes of Precision Anxiety in Orthopedic Manufacturing Partnerships
The Micron-Level Challenge in Implant Production
Orthopedic implants operate in an unforgiving environment where dimensional tolerances measured in microns directly impact clinical outcomes. A femoral stem with surface roughness exceeding specified parameters may compromise osseointegration, while a locking plate with hole position errors of even 0.1mm can prevent proper screw alignment during surgery. Medical device contract manufacturing partners must demonstrate not just capability to hit these tolerances occasionally, but statistical process control that ensures every production run maintains consistency. The anxiety emerges when OEMs discover that contract manufacturers who excel in general machining lack the specialized metrology equipment, environmental controls, and operator training required for medical-grade precision. Temperature fluctuations in the production environment, inadequate fixturing systems, or worn cutting tools create variation that falls within acceptable ranges for industrial components but represents catastrophic failure in orthopedic applications.
Documentation and Traceability Gaps
Regulatory bodies require complete material traceability from raw material certification through final inspection, yet many contract manufacturers approach documentation as an administrative burden rather than a quality imperative. Precision anxiety intensifies when OEMs audit their medical device contract manufacturing partners and discover gaps in batch records, missing calibration certificates, or inspection reports that lack the granularity required for FDA submissions. The challenge extends beyond simple record-keeping to encompass digital traceability systems that link every implant serial number to specific material lots, machine parameters, operator qualifications, and environmental conditions during production. Contract manufacturers accustomed to commercial manufacturing often underestimate the documentation rigor required, creating situations where physically acceptable parts cannot be released because the paper trail fails to meet regulatory standards. This disconnect between physical quality and documentary evidence generates significant anxiety for OEMs facing regulatory submissions with incomplete manufacturing records.
Capability Misrepresentation and Discovery Lag
Many orthopedic OEMs encounter precision anxiety after investing months in relationship development, only to discover during production validation that their contract manufacturing partner cannot consistently achieve the required specifications. This discovery lag occurs because initial capability assessments focus on best-case samples rather than statistical process capability over extended production runs. A medical device contract manufacturing partner may produce ten perfect prototype samples but lack the process stability to maintain those standards across thousands of units. The anxiety compounds when OEMs realize that switching suppliers mid-development creates significant delays, cost overruns, and potential regulatory complications. Contract manufacturers sometimes overstate their medical device experience, conflating general precision machining capabilities with the specialized requirements of implantable devices. This misalignment between represented and actual capabilities creates a trust deficit that permeates the entire partnership, forcing OEMs into intensive oversight roles that undermine the efficiency benefits of outsourcing.
Establishing Validation Frameworks That Build Confidence
Pre-Qualification Assessment Protocols
Effective management of precision anxiety begins long before purchase orders, with rigorous pre-qualification protocols that assess medical device contract manufacturing capabilities through objective evidence rather than marketing claims. Orthopedic OEMs should develop multi-dimensional assessment frameworks that evaluate not just current capabilities but organizational commitment to continuous improvement and regulatory compliance. This assessment includes on-site audits of metrology capabilities, review of quality management system documentation, evaluation of cleanroom or controlled environment facilities, and interviews with quality personnel to assess their understanding of medical device requirements. The most revealing assessment component involves requesting process capability studies for similar components, examining not just Cpk values but the underlying data collection methodology, measurement system analysis, and response to out-of-specification events. Contract manufacturers with genuine medical device contract manufacturing expertise readily provide this documentation and welcome detailed technical discussions, while those lacking depth become evasive or provide generic assurances without supporting data.
Graduated Production Validation Stages
Rather than immediately committing to full production volumes, sophisticated OEMs implement graduated validation stages that incrementally test their contract manufacturing partner's capabilities under increasingly realistic conditions. The first stage involves producing a small batch of the most challenging component features, with emphasis on dimensional verification using calibrated metrology equipment and multiple measurement techniques to eliminate instrument bias. This initial validation reveals whether the manufacturer's equipment, tooling strategies, and operator skills can consistently achieve the required tolerances. The second stage expands to complete assemblies or instrument sets, testing not just individual component precision but the manufacturer's ability to manage tolerance stack-ups, assembly sequences, and final inspection protocols. The final validation stage involves a full production run under normal manufacturing conditions, without the special attention that pilot runs often receive. This graduated approach allows OEMs to identify and address precision issues before committing to volumes that would make supplier changes prohibitively expensive, transforming medical device contract manufacturing relationships from leaps of faith into evidence-based partnerships.
Real-Time Quality Data Integration
Modern approaches to managing precision anxiety leverage digital quality systems that provide OEMs with real-time visibility into production quality metrics at their contract manufacturing partner's facility. These systems integrate data from coordinate measuring machines, surface roughness testers, and in-process inspection stations, automatically flagging deviations from statistical control limits before they result in non-conforming product. For medical device contract manufacturing relationships, this transparency transforms the traditional arms-length outsourcing model into a collaborative partnership where both parties share responsibility for quality outcomes. OEMs gain early warning of process drift that might indicate tool wear, material variation, or environmental changes, allowing proactive intervention rather than reactive correction after discovering defective batches. Contract manufacturers benefit from reduced customer audit frequency and increased trust, as continuous data transparency demonstrates their commitment to quality. Implementation requires upfront investment in compatible quality management systems and clear data-sharing protocols, but the resulting reduction in precision anxiety and improvement in first-pass yield justifies this investment for long-term partnerships focused on complex orthopedic devices.
Designing Collaborative Quality Agreements That Address Anxiety Triggers
Defining Mutual Quality Responsibilities
Precision anxiety often stems from ambiguity about which party bears responsibility for various quality activities in medical device contract manufacturing relationships. Effective quality agreements eliminate this ambiguity through explicit definition of responsibilities for incoming material inspection, in-process verification, final inspection protocols, non-conformance investigation, and corrective action implementation. The agreement should specify not just what activities occur but the acceptance criteria, measurement methods, sampling plans, and documentation requirements for each activity. For orthopedic devices, particular attention must address responsibilities for managing design changes, as even minor modifications can impact fit, function, and regulatory status. The quality agreement should establish clear protocols for change notification, impact assessment, validation requirements, and regulatory submission coordination. By documenting these responsibilities before production begins, OEMs and their contract manufacturing partners create shared expectations that reduce the likelihood of misunderstandings that generate anxiety during execution.
Establishing Escalation and Response Protocols
Even with robust processes, deviations and quality issues inevitably arise in medical device contract manufacturing, making escalation protocols essential for managing precision anxiety. Quality agreements should define specific trigger points that require immediate notification to the OEM, such as any out-of-specification measurement on critical dimensions, equipment failures affecting sterile barrier systems, or personnel deviations from validated procedures. The protocol should specify response timeframes, investigation depth requirements, and approval authorities for disposition decisions. For orthopedic implants where patient safety is paramount, the escalation protocol might require that no product ships until the OEM's quality team reviews and approves the investigation and corrective action. These protocols prevent situations where contract manufacturers make independent decisions about acceptability that later create regulatory complications or field performance issues. The existence of clear escalation paths actually reduces anxiety by ensuring that OEMs maintain appropriate oversight without needing to micromanage daily operations, allowing the medical device contract manufacturing relationship to balance efficiency with control.
Performance Metrics and Continuous Improvement Commitments
Quality agreements should establish quantitative performance metrics that allow both parties to objectively assess the health of the medical device contract manufacturing relationship and identify improvement opportunities before small issues escalate into major problems. Key metrics for orthopedic manufacturing include first-pass yield rates, dimensional capability indices for critical features, on-time delivery performance, documentation completeness scores, and customer complaint rates. The agreement should specify target values, measurement methodologies, reporting frequency, and review processes for these metrics. Beyond passive monitoring, effective agreements incorporate continuous improvement commitments where the contract manufacturer agrees to specific capability enhancement activities such as advanced metrology equipment investments, operator certification programs, or process automation projects. These commitments demonstrate the manufacturer's long-term commitment to the partnership and provide tangible evidence that they view medical device contract manufacturing as a strategic capability rather than just another customer segment. For OEMs, reviewing quarterly performance metrics against agreed targets provides reassurance that precision standards remain under control, directly addressing the underlying anxiety about consistency and reliability.
Implementing Risk Mitigation Strategies for High-Stakes Components
Dual-Source Qualification for Critical Components
While single-source medical device contract manufacturing relationships offer simplicity and potentially better pricing, they create significant vulnerability when precision issues arise or supply disruptions occur. Forward-thinking orthopedic OEMs mitigate precision anxiety by maintaining qualified secondary sources for their most critical components, particularly complex implants or specialized instruments that require extended lead times for supplier qualification. Dual-source strategies do not necessarily mean splitting production volume equally, but rather maintaining a qualified alternate supplier that can rapidly scale production if the primary manufacturer experiences quality issues, capacity constraints, or business continuity challenges. The qualification investment for secondary sources pays dividends in reduced anxiety, as OEMs maintain leverage in quality discussions and possess concrete alternatives if precision standards cannot be maintained. The secondary source also creates competitive pressure that often improves primary supplier performance, as contract manufacturers recognize they cannot take the business relationship for granted. For the most critical components in orthopedic systems, some OEMs maintain internal manufacturing capability alongside external contract manufacturing, providing ultimate control over precision and supply security while benefiting from external capacity and cost efficiency for the majority of production.
Advanced Process Monitoring and Predictive Maintenance
Precision degradation rarely occurs suddenly, typically resulting from gradual tool wear, calibration drift, or environmental changes that accumulate over time. Sophisticated medical device contract manufacturing partners implement advanced process monitoring systems that detect these early warning signals before they result in out-of-specification product. These systems might include real-time spindle vibration monitoring that predicts cutting tool failure, statistical process control charts that identify systematic drift in dimensional measurements, or environmental monitoring systems that correlate temperature and humidity changes with dimensional variation. Predictive maintenance programs use this data to schedule tool changes, equipment calibration, and preventive maintenance based on actual condition rather than arbitrary time intervals, maximizing equipment uptime while minimizing the risk of precision loss. For OEMs, partnering with contract manufacturers who invest in these advanced monitoring capabilities provides significant anxiety reduction, as data-driven maintenance prevents the surprise quality issues that undermine confidence. During supplier selection, OEMs should specifically assess the sophistication of process monitoring systems and the manufacturer's approach to predictive maintenance as indicators of their commitment to sustained precision in medical device contract manufacturing.
Regulatory Contingency Planning
A significant component of precision anxiety involves regulatory risk, specifically the concern that quality issues at a contract manufacturer could delay regulatory submissions, trigger warning letters, or necessitate field actions that damage brand reputation. Proactive OEMs address this anxiety through regulatory contingency planning that prepares responses for various failure scenarios. This planning includes pre-drafted change control procedures for supplier switches, template protocols for expanded inspection or testing, and communication strategies for regulatory agencies if issues arise. The contingency planning process should involve both the OEM's regulatory affairs team and the contract manufacturer's quality leadership, ensuring shared understanding of regulatory obligations and response authorities. For medical device contract manufacturing relationships supporting products in multiple global markets, contingency plans must address varying regulatory requirements and notification obligations across jurisdictions. While developing these contingency plans requires significant upfront effort, the planning process itself often identifies potential weaknesses in quality systems or supplier capabilities that can be addressed proactively, preventing the very scenarios the contingencies address. The existence of well-developed contingency plans paradoxically reduces anxiety by providing confidence that even worst-case scenarios have been anticipated and planned for, allowing OEM leadership to focus on strategic growth rather than operational worries.
Building Long-Term Partnerships That Transform Anxiety into Advantage
Investing in Supplier Capability Development
The most successful medical device contract manufacturing relationships evolve beyond transactional procurement into strategic partnerships where OEMs actively invest in their suppliers' capability development. This investment might include co-funding advanced metrology equipment specifically calibrated for the OEM's components, sponsoring operator training programs in medical device quality requirements, or providing technical expertise to optimize manufacturing processes for improved precision and yield. These investments create mutual commitment that transforms the relationship from easily substitutable vendor to strategic partner with specialized capabilities that would be difficult to replicate. For contract manufacturers, OEM investment in their capabilities enables them to pursue additional medical device business with enhanced credentials and equipment. For OEMs, these investments reduce precision anxiety by directly addressing capability gaps rather than simply hoping the supplier will improve independently. The investment approach works best when structured as capability development rather than charity, with clear performance expectations and metrics that demonstrate return on investment. Leading orthopedic OEMs view their key medical device contract manufacturing partners as extensions of their own operations, making strategic investments in supplier capabilities just as they would in internal manufacturing capacity.
Collaborative Design for Manufacturability
Many precision challenges in medical device contract manufacturing stem from designs developed without adequate consideration of manufacturing realities, creating tolerance requirements that push the boundaries of practical capability or specify features that require unnecessarily complex manufacturing processes. Progressive OEMs engage their contract manufacturing partners early in the design process, leveraging their manufacturing expertise to optimize designs for precision, cost, and reliability before finalizing specifications and initiating regulatory submissions. This collaborative approach might identify opportunities to adjust tolerances on non-critical features, modify geometry to improve machining access, or specify alternative materials that offer better dimensional stability. The design for manufacturability process reduces precision anxiety by ensuring that specifications align with demonstrated manufacturing capabilities rather than theoretical ideals. Contract manufacturers appreciate early involvement as it demonstrates respect for their expertise and creates shared ownership of product success. The collaborative design process also builds technical relationships between engineering teams that facilitate problem-solving when issues arise during production, as mutual respect and communication channels already exist. For medical device contract manufacturing relationships focused on complex orthopedic implants or instruments, design collaboration represents one of the highest-value activities for reducing precision anxiety while simultaneously improving product performance and manufacturability.
Transparency Through Integrated Quality Systems
The ultimate evolution of medical device contract manufacturing relationships involves integrated quality management systems that provide seamless data flow and shared visibility across organizational boundaries. In these advanced partnerships, the contract manufacturer's quality system automatically populates the OEM's quality database with inspection results, certificates of analysis, and process validation data, eliminating manual data transfer and reducing the documentation delays that create anxiety around regulatory submissions. Integrated systems enable OEMs to monitor key quality metrics in real-time dashboards, review inspection images remotely, and receive automated alerts when statistical control limits are approached. This transparency fundamentally changes the nature of the relationship from one based on periodic audits and inspection sampling to continuous monitoring and collaborative process control. Implementation requires significant investment in compatible information systems, data security protocols, and change management to align organizational processes, but the resulting reduction in precision anxiety and improvement in response speed justifies this investment for strategic partnerships. Contract manufacturers who embrace quality system integration demonstrate their commitment to transparency and quality, differentiating themselves from competitors who view quality data as proprietary information to be guarded. For orthopedic OEMs managing multiple complex medical device contract manufacturing relationships, integrated quality systems provide scalable oversight that maintains appropriate control without overwhelming internal quality resources.
FAQ
What are the most common sources of precision failures in medical device contract manufacturing for orthopedic implants?
The most frequent precision failures stem from inadequate process validation, insufficient environmental controls, and measurement system inadequacies rather than simple operator error. Many contract manufacturers underestimate the impact of temperature variation on dimensional stability, particularly for tight-tolerance features on large implants where thermal expansion significantly affects measurements. Another common issue involves measurement system capability, where inspection equipment lacks the resolution or repeatability required to reliably verify tolerances in the micron range, leading to false acceptance of out-of-specification parts or unnecessary rejection of conforming product. Process parameter drift represents a third major source, occurring when initial setup achieves required precision but gradual tool wear, fixture degradation, or material variation causes dimensions to drift over production runs. Addressing these common sources requires contract manufacturers to implement rigorous process validation protocols, invest in climate-controlled manufacturing environments, deploy high-resolution metrology equipment with regular calibration, and establish statistical process control systems that detect drift before it results in non-conforming product.
How can orthopedic OEMs assess whether a potential contract manufacturer truly understands medical device quality requirements?
Beyond reviewing certifications and marketing materials, OEMs should conduct technical interviews with the contract manufacturer's quality leadership, asking specific questions about their approach to design validation, process validation, and continued process verification as defined in FDA guidance documents. Genuine medical device contract manufacturing expertise reveals itself through detailed discussion of risk management processes, design history file documentation practices, and specific examples of how they have addressed validation challenges in previous projects. OEMs should request examples of actual validation protocols and reports, examining the rigor of acceptance criteria, statistical analysis methods, and investigation depth for any anomalies encountered during validation. Another revealing assessment involves touring the manufacturing facility and observing whether quality practices match stated procedures, including whether operators follow documented work instructions, environmental monitoring occurs as scheduled, and metrology equipment shows evidence of regular calibration and maintenance. Contract manufacturers with deep medical device experience welcome these detailed assessments and provide transparent access to systems and documentation, while those lacking genuine expertise become defensive or provide vague assurances without supporting evidence.
What contractual protections should orthopedic OEMs include to address precision anxiety in manufacturing agreements?
Effective manufacturing agreements should include specific performance guarantees around dimensional capability indices, with remedies if the contract manufacturer fails to maintain agreed capability levels over sustained production. These agreements should define clear acceptance criteria for initial production validation, including minimum sample sizes, statistical requirements, and OEM approval rights before full production authorization. The contract should address intellectual property protection for proprietary designs and manufacturing processes, particularly when the OEM provides specialized tooling or process development support. Liability provisions should clearly allocate responsibility for costs associated with quality failures, including rework, scrap, regulatory notification expenses, and potential field actions if defective product reaches the market. The agreement should grant the OEM comprehensive audit rights including unannounced inspections, with requirements that the contract manufacturer maintain records for specified retention periods and provide complete documentation upon request. For medical device contract manufacturing relationships, agreements should include provisions for business continuity, requiring the manufacturer to maintain adequate capacity, qualify backup equipment, and provide advance notice of any changes to facilities, key personnel, or subcontractors that might impact quality or supply continuity.
How do leading orthopedic OEMs balance cost pressure with the need for precision in contract manufacturing relationships?
Sophisticated OEMs recognize that precision in medical device contract manufacturing represents a non-negotiable requirement rather than an optional premium feature, shifting cost optimization efforts to areas that do not compromise quality. This approach involves rigorous design for manufacturability to eliminate unnecessarily tight tolerances on non-critical features, allowing cost reduction through relaxed specifications where clinical performance permits. OEMs achieve cost efficiency through long-term volume commitments that allow contract manufacturers to invest in dedicated tooling and process optimization, amortizing setup costs across larger production runs. Another strategy involves collaborative cost reduction initiatives where OEM and contract manufacturer engineering teams jointly identify process improvements, material optimizations, or automation opportunities that reduce cost while maintaining or improving precision. Leading OEMs avoid the false economy of selecting contract manufacturers solely on lowest quoted price, recognizing that precision failures generate far greater costs through rework, delays, regulatory complications, and potential field actions than the savings achieved through aggressive pricing. Instead, they evaluate total cost of ownership including quality performance, on-time delivery reliability, and responsiveness to issues, selecting medical device contract manufacturing partners who demonstrate sustainable cost structures supported by efficient processes rather than those who win business through unsustainably low pricing that inevitably leads to quality compromises.
Table of Contents
- Understanding the Root Causes of Precision Anxiety in Orthopedic Manufacturing Partnerships
- Establishing Validation Frameworks That Build Confidence
- Designing Collaborative Quality Agreements That Address Anxiety Triggers
- Implementing Risk Mitigation Strategies for High-Stakes Components
- Building Long-Term Partnerships That Transform Anxiety into Advantage
-
FAQ
- What are the most common sources of precision failures in medical device contract manufacturing for orthopedic implants?
- How can orthopedic OEMs assess whether a potential contract manufacturer truly understands medical device quality requirements?
- What contractual protections should orthopedic OEMs include to address precision anxiety in manufacturing agreements?
- How do leading orthopedic OEMs balance cost pressure with the need for precision in contract manufacturing relationships?

