orthopedic odm services
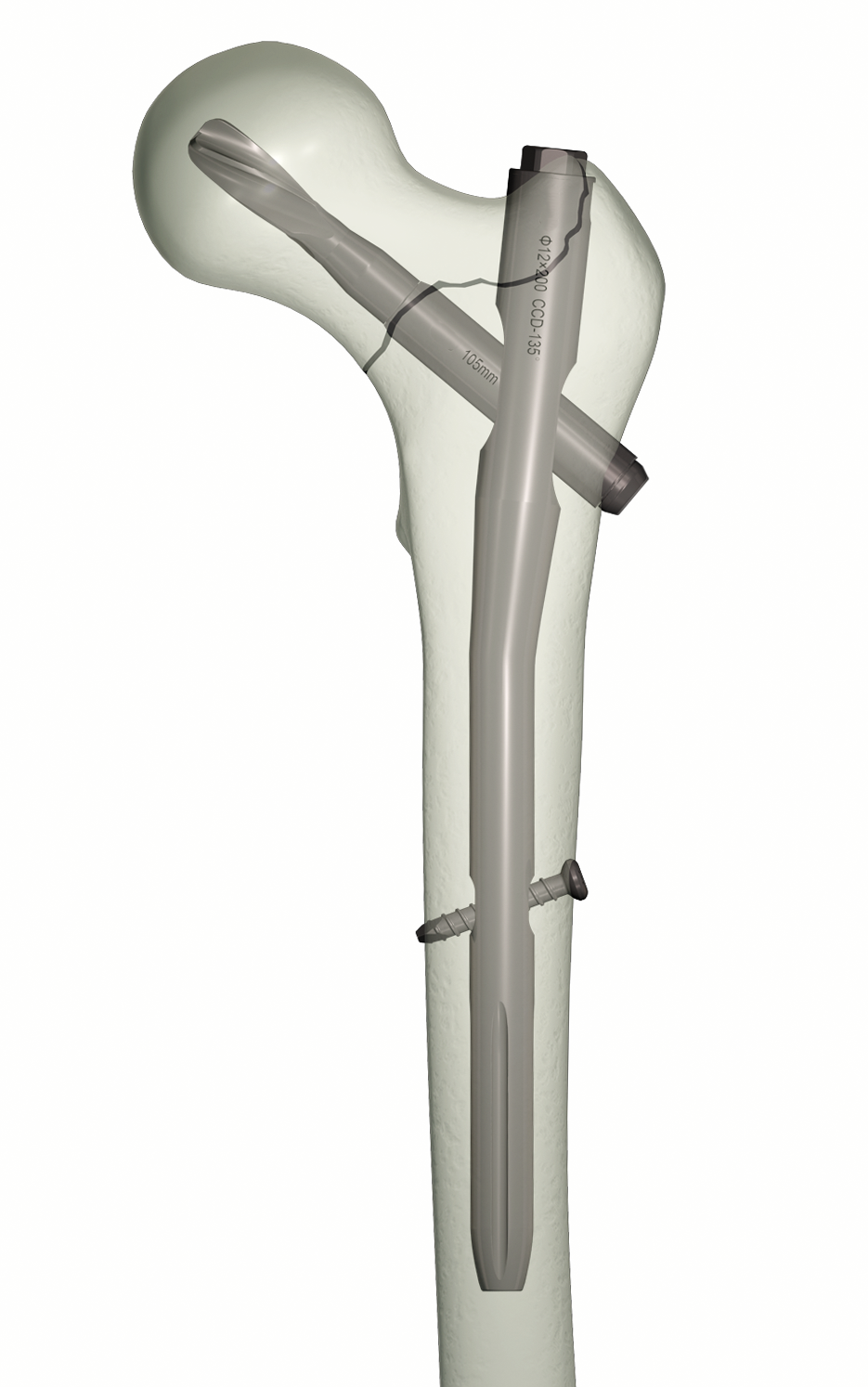
Orthopedic ODM services represent a comprehensive solution for medical device manufacturers seeking specialized production capabilities in the orthopedic sector. These services encompass the complete development and manufacturing process of orthopedic devices, from initial concept and design to final production. The service utilizes advanced manufacturing technologies, including precision CNC machining, 3D printing, and computer-aided design systems, to create high-quality orthopedic implants, instruments, and devices. State-of-the-art quality control measures ensure compliance with international medical device standards and regulations. The manufacturing facilities are equipped with clean rooms and advanced testing laboratories to maintain the highest levels of product quality and safety. These services cater to a wide range of orthopedic applications, including joint replacements, spinal devices, trauma fixation systems, and specialized surgical instruments. The integration of modern manufacturing techniques with experienced engineering teams allows for rapid prototyping and efficient scale-up to full production. Each project benefits from comprehensive documentation and validation processes, ensuring traceability and regulatory compliance throughout the manufacturing cycle.