Precision manufacturing serves as the cornerstone of successful orthopedic device development, particularly when partnering with an original equipment manufacturer. For medical device companies entering the orthopedic space, understanding the complete journey from initial concept through clinical implementation requires navigating complex regulatory landscapes, mastering material science principles, and ensuring manufacturing precision that meets the exacting standards of surgical applications. The orthopedic device OEM relationship transforms innovative ideas into tangible clinical solutions that improve patient outcomes while maintaining compliance with international quality standards.
The orthopedic medical device industry demands unprecedented levels of precision, where tolerances measured in microns directly impact surgical success and patient safety. Selecting the right manufacturing partner for your orthopedic device concept involves evaluating technical capabilities, quality management systems, regulatory expertise, and the ability to translate clinical requirements into manufacturable designs. This comprehensive guide walks through each critical phase of the orthopedic device OEM process, providing actionable insights for companies developing joint replacement systems, trauma fixation devices, spinal implants, surgical instruments, and specialized orthopedic tools.
Understanding the Orthopedic Device OEM Landscape
Defining the Role of Manufacturing Partners in Medical Device Development
An orthopedic device OEM functions as a specialized manufacturing partner that bridges the gap between clinical innovation and market-ready medical products. These manufacturers possess the technical infrastructure, regulatory knowledge, and quality systems necessary to transform conceptual designs into FDA-cleared or CE-marked orthopedic devices. Unlike general contract manufacturers, orthopedic-focused OEM partners understand the unique biomechanical requirements, biocompatibility standards, and sterilization protocols specific to implantable and surgical devices. Their expertise spans material selection, precision machining, surface finishing, and validation testing that ensures devices perform reliably within the demanding physiological environment of the human musculoskeletal system.
The value proposition of partnering with an established orthopedic device OEM extends beyond manufacturing capacity. These specialized partners contribute design-for-manufacturability insights during product development, helping avoid costly redesigns by identifying potential production challenges early in the concept phase. They maintain relationships with material suppliers certified for medical applications, operate cleanroom facilities meeting ISO Class 7 or Class 8 standards, and employ quality engineers trained in risk management methodologies specific to medical devices. For emerging medical device companies, this partnership model provides access to capital-intensive infrastructure without requiring direct investment in specialized equipment, allowing faster market entry and reduced financial risk during product commercialization.
Key Capabilities That Distinguish Orthopedic Manufacturing Excellence
Superior orthopedic device OEM partners demonstrate mastery across multiple technical domains that directly influence device performance and regulatory compliance. Precision machining capabilities represent a fundamental requirement, with multi-axis CNC systems capable of achieving tolerances within plus-or-minus five microns on critical features such as bearing surfaces, screw threads, and locking mechanisms. Material expertise extends to understanding the mechanical properties, corrosion resistance, and biological response characteristics of titanium alloys, cobalt-chromium systems, stainless steel grades, and advanced polymers like ultra-high-molecular-weight polyethylene. Surface engineering knowledge encompasses electropolishing, passivation, plasma coating, and texture control that influences osseointegration, wear resistance, and bacterial adhesion.
Beyond technical manufacturing skills, leading orthopedic device OEM providers maintain comprehensive quality management systems aligned with ISO 13485 standards and FDA 21 CFR Part 820 requirements. These systems govern every aspect of production, from incoming material inspection through final device release, with documented procedures for process validation, equipment calibration, environmental monitoring, and corrective action implementation. Advanced partners invest in metrology equipment including coordinate measuring machines, optical profilometers, and non-destructive testing systems that verify dimensional accuracy, surface finish, and internal integrity. Their validation protocols address installation qualification, operational qualification, and performance qualification for all manufacturing processes, creating the documented evidence required for regulatory submissions and ongoing compliance maintenance.
Industry Segments and Application Categories
The orthopedic device OEM sector serves diverse market segments, each presenting unique technical requirements and regulatory considerations. Joint reconstruction devices, including hip and knee replacement systems, demand exceptional precision in bearing surface geometry and material compatibility to minimize wear debris generation over decades of service. Trauma fixation products such as bone plates, intramedullary nails, and external fixation systems require designs that balance mechanical strength with anatomical adaptation, often incorporating variable-angle locking technology and minimally invasive delivery systems. Spinal implants present challenges related to complex three-dimensional geometries, porous coating integration for biological fixation, and instrument systems that enable accurate surgical placement through small incisions.
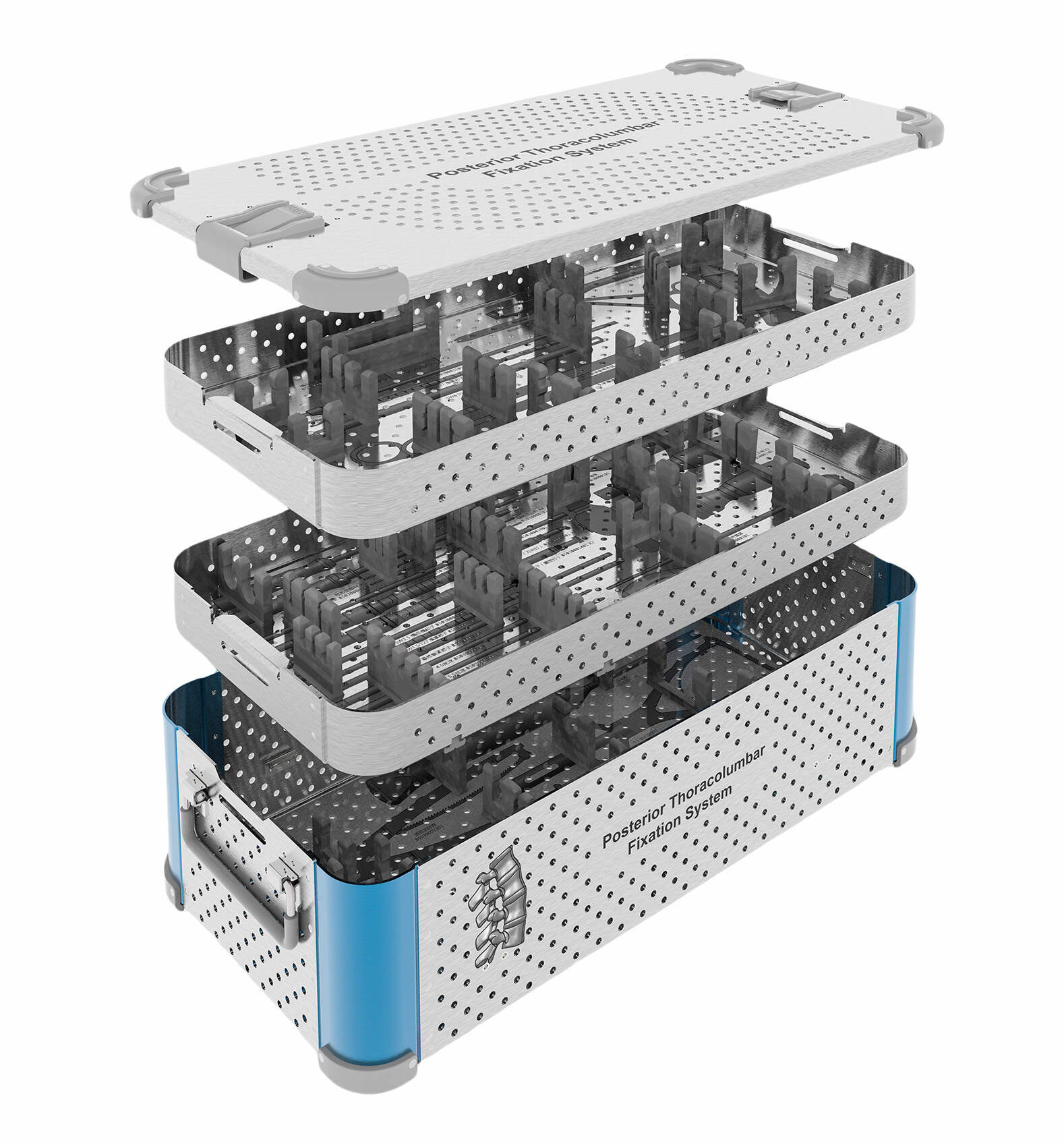
Surgical instrument manufacturing within the orthopedic device OEM framework encompasses specialized tools designed for bone preparation, implant insertion, and intraoperative measurement. These instruments must withstand repeated sterilization cycles while maintaining dimensional stability and functional performance. Storage and organization systems, including orthopedic device OEM instrument cases, play critical roles in surgical efficiency and contamination prevention. Custom procedure trays and modular case systems organize surgical sets according to procedural workflow, protecting delicate instruments during transport and sterilization while facilitating efficient setup in the operating room. The design and manufacturing of these accessory systems require understanding surgical protocols, sterilization compatibility, and healthcare facility workflow optimization.
Navigating the Product Development Pathway
Conceptualization and Clinical Needs Assessment
Successful orthopedic device development begins with thorough clinical needs assessment that identifies specific unmet requirements within surgical practice. This foundational phase involves collaboration between surgeons, biomedical engineers, and the orthopedic device OEM partner to define performance criteria, anatomical fit requirements, and clinical outcome objectives. Market research informs decisions regarding target patient populations, competitive differentiation strategies, and reimbursement landscape considerations that influence design priorities. Understanding the clinical workflow where the device will function helps establish usability requirements, procedural time constraints, and integration considerations with existing surgical techniques and complementary instruments.
During concept development, the orthopedic device OEM contributes manufacturability analysis that shapes design decisions before significant engineering resources are committed. This early engagement identifies material options that balance clinical performance requirements with production feasibility, surface finish specifications that can be reliably achieved and verified, and geometric features that align with available manufacturing processes. Risk assessment during this phase addresses potential failure modes, biocompatibility concerns, and regulatory classification implications. Establishing clear design inputs that translate clinical needs into measurable engineering specifications creates the foundation for systematic development, reducing the likelihood of costly iterations during later validation phases.
Design Development and Engineering Verification
The design development phase transforms conceptual requirements into detailed engineering specifications suitable for manufacturing implementation. Computer-aided design systems enable creation of three-dimensional models that capture complex anatomical geometries, interface features, and assembly relationships. Finite element analysis evaluates mechanical performance under physiological loading conditions, predicting stress distributions, fatigue life, and potential failure locations. The orthopedic device OEM manufacturing partner reviews designs for producibility, suggesting modifications that enhance manufacturing efficiency without compromising clinical performance. Design for manufacturability considerations include tool access for machining operations, fixturing strategies for multi-sided processing, and inspection methods for critical dimensions.
Engineering verification testing validates that the device design meets specified requirements through objective measurement and analysis. Mechanical testing protocols evaluate ultimate strength, yield properties, fatigue endurance, and wear resistance using standardized test methods from organizations like ASTM International and ISO technical committees. Dimensional verification employs precision metrology to confirm that manufactured prototypes conform to drawing specifications within established tolerances. Material characterization testing verifies chemical composition, microstructure, and mechanical properties of raw materials and finished components. The orthopedic device OEM facility conducts these verification activities within controlled environments using calibrated equipment and documented procedures, generating the objective evidence required for design history files and regulatory submissions.
Prototype Development and Iterative Refinement
Prototype manufacturing represents a critical transition from theoretical design to physical realization, revealing practical challenges that may not be apparent in digital models. Initial prototypes produced by the orthopedic device OEM partner serve multiple purposes, including design verification testing, surgical technique development, and clinical evaluation planning. Rapid prototyping technologies such as additive manufacturing enable quick iteration of complex geometries, allowing surgeons to evaluate anatomical fit and instrument ergonomics before committing to production tooling. However, prototypes intended for mechanical testing or clinical use typically require manufacturing processes identical to planned production methods, ensuring that material properties and dimensional characteristics accurately represent the final device.
Iterative refinement based on prototype evaluation addresses both clinical performance and manufacturing efficiency considerations. Surgeon feedback during simulated or cadaveric procedures identifies usability improvements, visualization enhancements, and ergonomic adjustments that optimize surgical workflow. The orthopedic device OEM manufacturing team contributes insights regarding process capability, yield optimization, and cost reduction opportunities that maintain device performance while improving production economics. This collaborative refinement process continues until the design achieves stable performance across all critical characteristics, demonstrating readiness for formal design validation and process validation activities that precede commercial manufacturing.
Manufacturing Process Implementation and Quality Assurance
Material Selection and Supply Chain Management
Material selection fundamentally influences orthopedic device performance, biocompatibility, and manufacturing feasibility. The orthopedic device OEM partner maintains qualified supplier relationships for medical-grade materials that meet stringent composition, purity, and traceability requirements. Titanium alloys, particularly Ti-6Al-4V, offer excellent biocompatibility, corrosion resistance, and strength-to-weight ratios suitable for implantable devices, while cobalt-chromium alloys provide superior wear resistance for bearing surfaces in joint replacement applications. Stainless steel grades such as 316L serve trauma fixation applications where temporary implantation or instrument fabrication requires cost-effective materials with adequate mechanical properties and corrosion resistance.
Supply chain management for orthopedic device manufacturing requires rigorous material traceability from mill certification through finished device labeling. Each material lot undergoes incoming inspection that verifies chemical composition, mechanical properties, and surface condition against purchase specifications. The orthopedic device OEM maintains approved supplier lists based on audit results, quality history, and regulatory compliance status. Material certifications documenting heat treatment, test results, and conformance to specifications become part of the device history record, enabling complete traceability for post-market surveillance and regulatory inspections. Advanced partners implement vendor-managed inventory systems that ensure material availability while minimizing carrying costs and maintaining lot-level traceability throughout the production cycle.
Precision Machining and Manufacturing Technologies
Precision machining forms the backbone of orthopedic device manufacturing, transforming raw material into components meeting exacting dimensional and surface finish requirements. Multi-axis CNC milling centers equipped with high-speed spindles and advanced tooling systems execute complex three-dimensional geometries with repeatability measured in single-digit microns. Swiss-type turning centers produce small-diameter components such as bone screws and cannulated instruments with exceptional concentricity and surface finish. The orthopedic device OEM invests in machine tools specifically configured for medical device production, including sealed work zones that minimize particulate contamination and automation systems that reduce human handling during critical manufacturing steps.
Advanced manufacturing technologies complement traditional machining processes to achieve specialized device features and properties. Wire electrical discharge machining creates intricate geometries and sharp internal corners impossible to produce with rotating cutting tools. Laser marking systems apply permanent, high-contrast identification codes without generating particulate debris or altering surface chemistry. Additive manufacturing technologies, including electron beam melting and direct metal laser sintering, enable production of porous structures that promote biological fixation and patient-specific anatomical adaptations. The orthopedic device OEM integrates these diverse technologies within validated manufacturing processes, maintaining documented procedures for setup, operation, and verification that ensure consistent output meeting design specifications.
Surface Treatment and Finishing Operations
Surface characteristics profoundly influence orthopedic device performance, affecting osseointegration, wear resistance, corrosion behavior, and bacterial adhesion. The orthopedic device OEM implements specialized surface treatment processes tailored to device function and material composition. Electropolishing removes surface irregularities from stainless steel and titanium components while enhancing corrosion resistance through passive film development. Plasma spray coating applies titanium or hydroxyapatite layers that promote bone ingrowth on press-fit implant surfaces. Anodization creates controlled oxide layers on titanium devices, enabling color-coding for size identification while maintaining biocompatibility.
Surface finish specifications for orthopedic devices range from highly polished bearing surfaces with roughness values below 0.05 micrometers to textured bone-contacting regions optimized for biological fixation. Achieving and verifying these surfaces requires sophisticated metrology equipment including optical profilometers, scanning electron microscopes, and atomic force microscopes. The orthopedic device OEM establishes validated cleaning processes that remove machining residues, polishing compounds, and organic contaminants without introducing new contamination or altering surface properties. Final passivation treatments enhance corrosion resistance on stainless steel components while maintaining dimensional stability and surface cleanliness required for sterilization and long-term implantation.
Quality Control and Inspection Protocols
Comprehensive quality control systems distinguish leading orthopedic device OEM manufacturers, ensuring every device meets specifications before release. Statistical process control monitors critical manufacturing parameters in real-time, detecting process drift before nonconforming product is generated. First-article inspection verifies that initial production units from new setups or process changes conform to all dimensional, material, and functional requirements. In-process inspection at strategic manufacturing stages prevents defective components from advancing through subsequent operations, reducing scrap costs and identifying process improvement opportunities.
Final inspection protocols combine automated measurement systems with skilled manual inspection to verify all critical device characteristics. Coordinate measuring machines evaluate complex three-dimensional geometries with sub-micron accuracy, comparing manufactured features against CAD models. Optical comparators and vision systems inspect surface finish, edge conditions, and marking legibility. Functional testing validates assembly torques, locking mechanisms, and instrument articulation through documented test procedures. The orthopedic device OEM maintains calibration programs for all measurement equipment, with traceability to national standards and recalibration intervals based on equipment type, usage frequency, and measurement criticality. Inspection records become part of the device history record, documenting conformance for regulatory compliance and enabling root cause analysis if field issues arise.
Regulatory Compliance and Clinical Validation
Understanding Medical Device Classification and Regulatory Pathways
Regulatory classification fundamentally shapes the development timeline, validation requirements, and market entry strategy for orthopedic devices. Most orthopedic implants fall under Class II or Class III designation in the United States, requiring either 510(k) premarket notification or premarket approval based on risk profile and predicate device availability. The orthopedic device OEM partner contributes regulatory intelligence regarding classification rationale, predicate device identification, and substantial equivalence arguments that support efficient regulatory submissions. Understanding European Medical Device Regulation and other international frameworks enables coordinated development strategies that address multiple markets simultaneously, reducing overall time to commercial availability.
Regulatory pathway selection influences design control documentation, validation testing scope, and clinical data requirements. Devices with established predicates may proceed through abbreviated regulatory routes, while novel technologies require comprehensive safety and effectiveness demonstration through clinical studies. The orthopedic device OEM manufacturing partner supports regulatory submissions by providing manufacturing process descriptions, validation protocols, quality system documentation, and facility registration information. Their experience with regulatory inspections and audit responses helps anticipate reviewer questions and prepare comprehensive responses that expedite approval processes. Maintaining current registrations with FDA, notified bodies, and international regulators demonstrates the OEM's commitment to compliance and reduces risk for device developers relying on their manufacturing capabilities.
Biocompatibility Testing and Material Characterization
Biocompatibility evaluation represents a critical validation requirement for orthopedic devices contacting tissue or bone during clinical use. ISO 10993 standards define a systematic approach to biological safety assessment based on device characteristics, contact duration, and tissue type. The orthopedic device OEM coordinates with accredited testing laboratories to conduct required biocompatibility studies, including cytotoxicity, sensitization, irritation, systemic toxicity, and implantation testing for materials and final devices. Testing protocols account for sterilization effects, leachable substances, and worst-case exposure scenarios that represent clinical use conditions.
Material characterization extends beyond basic biocompatibility to address specific performance requirements of orthopedic applications. Corrosion testing evaluates electrochemical behavior in physiological environments, assessing both general corrosion rates and susceptibility to localized attack such as pitting or crevice corrosion. Wear testing for bearing surfaces employs joint simulators that replicate physiological motion patterns and loading cycles, quantifying debris generation rates and dimensional changes over millions of cycles. The orthopedic device OEM conducts these specialized tests within facilities equipped for long-duration studies, maintaining environmental controls and employing validated analytical methods that generate defensible data for regulatory submissions and scientific publication.
Sterilization Validation and Packaging Development
Sterilization validation ensures that orthopedic devices reach clinical users in a sterile state while maintaining functional performance and material properties. The orthopedic device OEM evaluates sterilization modality options including steam autoclaving, ethylene oxide, gamma irradiation, and electron beam processing based on material compatibility, geometric complexity, and packaging requirements. Validation protocols following ISO 11135, ISO 11137, or ISO 17665 standards demonstrate that the chosen sterilization process achieves the required sterility assurance level without causing device degradation or introducing harmful residuals.
Packaging design for sterile orthopedic devices balances protective requirements with sterilant penetration, shelf life stability, and ease of aseptic presentation in the operating room. The orthopedic device OEM develops packaging systems using medical-grade materials such as Tyvek pouches, thermoformed trays, and rigid containers that maintain sterile barriers while withstanding distribution stresses. Package validation testing demonstrates seal integrity, sterile barrier performance, and stability throughout claimed shelf life under specified storage conditions. Transit testing protocols subject packaged devices to vibration, compression, and drop scenarios representing transportation hazards, verifying that package integrity and device condition remain acceptable after distribution to end users.
Clinical Evaluation and Post-Market Surveillance
Clinical validation demonstrates that orthopedic devices perform safely and effectively when used according to their intended purpose by trained healthcare providers. Clinical study design varies from literature-based evaluations leveraging published data on similar devices to prospective clinical trials enrolling patients and tracking outcomes over extended follow-up periods. The orthopedic device OEM supports clinical activities by providing manufacturing documentation, shelf life data, and technical support for investigator training. Their quality systems enable traceability of devices used in clinical studies, facilitating correlation of performance data with manufacturing records if device-related events occur.
Post-market surveillance systems maintain ongoing monitoring of device performance after commercial release, detecting rare adverse events and performance trends that inform continuous improvement initiatives. The orthopedic device OEM implements complaint handling procedures that document device-related issues, conduct investigations, and implement corrective actions when systematic problems are identified. Medical device reporting obligations require timely notification to regulatory authorities when serious injuries or malfunctions occur. Vigilance data analysis identifies opportunities for design improvements, manufacturing process refinements, or enhanced user training that optimize device performance and patient safety throughout the product lifecycle.
Strategic Considerations for OEM Partnership Success
Evaluating Manufacturing Capabilities and Technical Competencies
Selecting an orthopedic device OEM partner requires thorough assessment of technical capabilities aligned with specific device requirements. Facility tours reveal equipment sophistication, cleanliness standards, and organizational culture that influence manufacturing quality. Evaluation criteria should address machine tool accuracy and maintenance programs, metrology equipment calibration status, and environmental monitoring systems for cleanroom operations. The orthopedic device OEM should demonstrate process capability studies showing statistical control of critical manufacturing parameters with process capability indices exceeding 1.33 for key device characteristics. Quality system maturity assessment examines documentation systems, training programs, and continuous improvement initiatives that sustain compliance and performance excellence.
Technical competency evaluation extends beyond manufacturing equipment to encompass engineering expertise and problem-solving capability. The orthopedic device OEM team should include engineers with advanced degrees in mechanical engineering, materials science, or biomedical engineering who understand both clinical requirements and manufacturing constraints. Experience with similar device types, material systems, and regulatory pathways reduces development risk and accelerates time to market. References from existing clients provide insights into communication responsiveness, schedule reliability, and collaborative approach during challenging development phases. Financial stability assessment ensures the OEM partner possesses resources to support long-term production commitments and invest in technology upgrades that maintain manufacturing competitiveness.
Intellectual Property Protection and Confidentiality Management
Intellectual property considerations significantly influence orthopedic device OEM relationships, particularly for innovative technologies representing substantial competitive advantages. Comprehensive confidentiality agreements should precede detailed design disclosure, clearly defining proprietary information scope, permitted uses, and return or destruction obligations upon project completion. The orthopedic device OEM should maintain information security systems that prevent unauthorized access to design files, prevent cross-contamination between competing client projects, and implement employee training on confidentiality obligations. Physical and digital access controls, visitor management procedures, and secure communication protocols demonstrate commitment to protecting client intellectual property.
Ownership of improvements and manufacturing process innovations developed during the OEM relationship requires clear contractual definition. Agreements should specify whether design modifications suggested by the orthopedic device OEM vest with the device developer, the manufacturer, or create joint ownership requiring mutual consent for commercialization. Tooling ownership provisions address whether custom fixtures, molds, and inspection devices remain the property of the device developer or the OEM upon project termination. These provisions influence cost allocation during development and affect the device developer's ability to transfer manufacturing to alternative suppliers if business circumstances change. Experienced legal counsel specializing in medical device contracts ensures agreements balance protection of proprietary information with practical business flexibility.
Cost Management and Commercial Sustainability
Cost structures for orthopedic device manufacturing reflect complex interactions between material expenses, processing time, quality control requirements, and regulatory compliance overhead. The orthopedic device OEM should provide transparent cost breakdowns that enable informed decisions regarding design optimization and volume commitments. Non-recurring engineering costs cover development activities including prototype fabrication, process validation, and quality system documentation. Tooling investments encompass fixtures, cutting tools, and inspection equipment specific to the device. Per-unit production costs include material, labor, overhead allocation, and quality testing for each manufactured device. Understanding these cost components enables realistic pricing strategies and informs make-versus-buy decisions for device companies considering vertical integration.
Long-term commercial sustainability requires alignment between device developer growth expectations and the orthopedic device OEM capacity planning. Volume projections influence decisions regarding dedicated production lines, inventory management approaches, and supplier agreements for raw materials. Contractual provisions should address capacity reservation, minimum order quantities, and price adjustment mechanisms that account for volume changes, material cost fluctuations, and regulatory requirement evolution. Forecasting accuracy expectations and order lead time requirements establish operational parameters that balance inventory carrying costs against stockout risks. Collaborative business planning sessions between device developers and their orthopedic device OEM partners create shared understanding of market dynamics, enabling proactive capacity investments and cost optimization initiatives that sustain competitive positioning.
Building Collaborative Relationships and Communication Frameworks
Successful orthopedic device OEM partnerships transcend transactional supplier relationships to create collaborative alliances that leverage complementary expertise. Regular communication cadences including weekly production status updates, monthly quality reviews, and quarterly business planning sessions maintain alignment and enable proactive issue resolution. The orthopedic device OEM should assign dedicated project managers who serve as single points of contact, coordinating across engineering, quality, and production functions to provide cohesive support. Cross-functional teams including representatives from both organizations address complex technical challenges through structured problem-solving methodologies that generate innovative solutions benefiting device performance and manufacturing efficiency.
Conflict resolution mechanisms establish procedures for addressing disagreements regarding specifications, quality acceptance, or delivery schedules before they escalate into commercial disputes. Escalation paths define management levels empowered to make decisions at various disagreement severities, ensuring appropriate authority engagement without unnecessary delay. Performance metrics including on-time delivery rates, first-pass yield percentages, and customer complaint frequencies provide objective assessment of the orthopedic device OEM relationship health. Periodic relationship reviews conducted at executive levels evaluate strategic alignment, identify continuous improvement opportunities, and reinforce mutual commitment to partnership success. These governance structures create stable foundations for long-term collaboration that benefits both device innovation and commercial execution.
FAQ
What qualifications should an orthopedic device OEM possess to ensure regulatory compliance?
An qualified orthopedic device OEM must maintain ISO 13485 certification demonstrating conformance to medical device quality management system requirements, along with facility registration with relevant regulatory authorities such as FDA establishment registration in the United States. The manufacturer should have documented experience with design control processes per FDA 21 CFR Part 820 Subpart C, including formal design review procedures, verification and validation protocols, and design transfer processes. Evidence of successful regulatory submissions for similar device types, including 510(k) clearances or CE mark approvals, indicates familiarity with technical documentation requirements and reviewer expectations. Regular inspections by regulatory authorities with satisfactory outcomes demonstrate sustained compliance rather than one-time certification achievements. Additionally, the OEM should maintain quality agreements with critical suppliers and contract testing laboratories, ensuring the entire supply chain meets medical device quality standards.
How long does the typical development timeline span from concept to commercial manufacturing?
Development timelines for orthopedic devices vary significantly based on device complexity, regulatory classification, and clinical validation requirements, typically ranging from eighteen months to five years. Straightforward Class II devices with clear predicate pathways and limited novel features may progress from concept to FDA clearance within eighteen to thirty-six months, encompassing design development, verification testing, process validation, and regulatory submission preparation. Complex Class III devices requiring clinical trials to demonstrate safety and effectiveness typically require four to five years or longer, with clinical study design, site recruitment, patient enrollment, and follow-up data collection consuming substantial portions of the timeline. The orthopedic device OEM manufacturing partnership becomes critical during process development and validation phases, which typically span six to twelve months and include installation qualification, operational qualification, and performance qualification activities demonstrating consistent manufacturing capability. Parallel development activities including simultaneous engineering verification and process development can compress overall timelines, though regulatory submission preparation and authority review periods remain largely outside the control of development teams.
What cost factors most significantly influence orthopedic device manufacturing pricing?
Material costs represent substantial components of orthopedic device manufacturing expenses, particularly for implantable devices utilizing titanium alloys, cobalt-chromium systems, or specialized biocompatible polymers that command premium pricing due to medical-grade certifications and lot traceability requirements. Manufacturing complexity directly influences per-unit costs through processing time requirements, with intricate geometries demanding multi-axis machining operations consuming more machine hours and requiring specialized tooling compared to simpler designs. Quality control and inspection intensity scales with device criticality, with implantable devices requiring more extensive dimensional verification, material testing, and documentation than non-implantable surgical instruments, though both require rigorous controls. Surface treatment and finishing operations such as electropolishing, plasma coating, or anodization add significant cost depending on specification stringency and required surface characteristics. Regulatory compliance overhead including validation study execution, quality system maintenance, and documentation management distributes across production volumes, making low-volume specialty devices proportionally more expensive than high-volume standard products. The orthopedic device OEM pricing structure should transparently separate these components, enabling device developers to identify cost reduction opportunities through design optimization, material substitution evaluation, or volume commitment increases.
Can emerging companies access orthopedic device OEM partnerships without minimum volume commitments?
Many orthopedic device OEM manufacturers recognize the strategic value of supporting emerging companies developing innovative technologies, even when initial production volumes remain modest compared to established product lines. Flexible partnership models may include higher per-unit pricing for low-volume production that reflects setup costs and capacity allocation without requiring large minimum order quantities that exceed early-stage market demand. Some OEM partners operate dedicated innovation divisions or startup incubator programs specifically designed to support device developers through early commercialization phases, providing scaled pricing structures that adjust as volumes grow and products achieve market traction. Hybrid arrangements combining development services with manufacturing options enable device companies to progress through design finalization and process validation using OEM expertise, then commit to production agreements once regulatory clearance is achieved and commercial forecasts become more certain. Device developers should transparently communicate volume projections, growth expectations, and funding status during OEM selection, enabling manufacturers to propose partnership structures aligned with realistic business trajectories. While some large-scale orthopedic device OEM operations focus exclusively on high-volume established products, numerous specialty manufacturers actively cultivate relationships with innovative companies, viewing these partnerships as strategic investments in future growth opportunities and technology portfolio diversification.
Table of Contents
- Understanding the Orthopedic Device OEM Landscape
- Navigating the Product Development Pathway
- Manufacturing Process Implementation and Quality Assurance
- Regulatory Compliance and Clinical Validation
- Strategic Considerations for OEM Partnership Success
-
FAQ
- What qualifications should an orthopedic device OEM possess to ensure regulatory compliance?
- How long does the typical development timeline span from concept to commercial manufacturing?
- What cost factors most significantly influence orthopedic device manufacturing pricing?
- Can emerging companies access orthopedic device OEM partnerships without minimum volume commitments?

